Among the oxides of nitrogen ${{\text{N}}_{2}}{{\text{O}}_{3}}\text{, }{{\text{N}}_{2}}{{\text{O}}_{4}}\text{ and }{{\text{N}}_{2}}{{\text{O}}_{5}}$;molecule(s) having nitrogen-nitrogen bond is/are:
$\begin{align}
& \text{A}\text{. Only }{{\text{N}}_{2}}{{\text{O}}_{5}} \\
& \text{B}\text{. }{{\text{N}}_{2}}{{\text{O}}_{3}}\text{ and }{{\text{N}}_{2}}{{\text{O}}_{4}} \\
& \text{C}\text{. }{{\text{N}}_{2}}{{\text{O}}_{3}}\text{ }\text{and }{{\text{N}}_{2}}{{\text{O}}_{5}} \\
& \text{D}\text{. }{{\text{N}}_{2}}{{\text{O}}_{4}}\text{ and }{{\text{N}}_{2}}{{\text{O}}_{5}} \\
\end{align}$
Answer
585k+ views
Hint: Electronic configuration of the nitrogen is \[\text{1}{{\text{s}}^{2}}\text{ 2}{{\text{s}}^{2}}\text{ 2}{{\text{p}}^{3}}\] due to which it can form 3 bonds easily with other atoms. The electronegativity of oxygen is more than the nitrogen because of its larger size than nitrogen. Electronegativity is the tendency of an element to attract a pair of electrons from another element.
Complete Solution:
-We have to identify the N - N bond from the given three compounds of nitrogen oxide which are also known as acidic oxides.
-So, firstly we have to draw the structure of nitrogen trioxide, nitrogen tetroxide and nitrogen penta-oxide which have molecular formula of ${{\text{N}}_{2}}{{\text{O}}_{3}}\text{, }{{\text{N}}_{2}}{{\text{O}}_{4}}\text{ and }{{\text{N}}_{2}}{{\text{O}}_{5}}$ respectively.
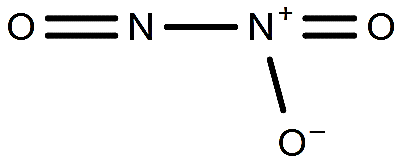
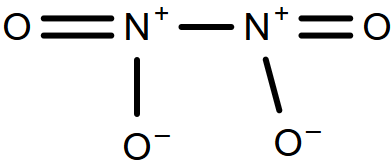
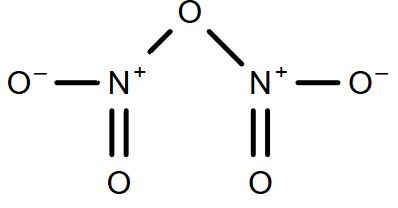
-So, we can see that only ${{\text{N}}_{2}}{{\text{O}}_{3}}$ and ${{\text{N}}_{2}}{{\text{O}}_{4}}$ have only one N-N bond.
-In ${{\text{N}}_{2}}{{\text{O}}_{5}}$, there is 3 N - O and 2 N = O bond and not N-N bond so it will be the incorrect answer.
-The name of the compound ${{\text{N}}_{2}}{{\text{O}}_{3}}$ is nitrogen trioxide and it is formed when the nitrogen oxide and nitrogen dioxide reacts together i.e. \[\text{NO + N}{{\text{O}}_{2}}\to \text{ }{{\text{N}}_{2}}{{\text{O}}_{3}}\].
-Whereas when two molecules of nitrogen oxide dimerise with each other they form nitrogen tetroxide i.e. \[2\text{N}{{\text{O}}_{2}}\ \rightleftharpoons \text{ }{{\text{N}}_{2}}{{\text{O}}_{4}}\]
-Moreover, the oxidation state of nitrogen in nitrogen trioxide, nitrogen tetroxide and nitrogen penta-oxide is +2, +3 and +5.
-So, only nitrogen trioxide and nitrogen tetroxide will have a N - N bond.
Note: Nitrogen can form maximum of 3 covalent bond and 1 co-ordinate bond because according to the electronic configuration it has ability to form 3 bonds easily but in some cases, it can also share some a pair electron i.e. lone pair which forms coordination bond.
Complete Solution:
-We have to identify the N - N bond from the given three compounds of nitrogen oxide which are also known as acidic oxides.
-So, firstly we have to draw the structure of nitrogen trioxide, nitrogen tetroxide and nitrogen penta-oxide which have molecular formula of ${{\text{N}}_{2}}{{\text{O}}_{3}}\text{, }{{\text{N}}_{2}}{{\text{O}}_{4}}\text{ and }{{\text{N}}_{2}}{{\text{O}}_{5}}$ respectively.
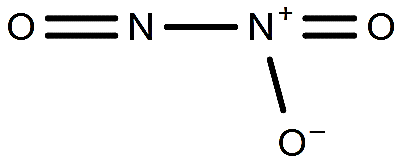
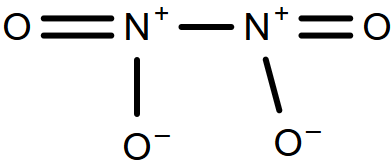

-So, we can see that only ${{\text{N}}_{2}}{{\text{O}}_{3}}$ and ${{\text{N}}_{2}}{{\text{O}}_{4}}$ have only one N-N bond.
-In ${{\text{N}}_{2}}{{\text{O}}_{5}}$, there is 3 N - O and 2 N = O bond and not N-N bond so it will be the incorrect answer.
-The name of the compound ${{\text{N}}_{2}}{{\text{O}}_{3}}$ is nitrogen trioxide and it is formed when the nitrogen oxide and nitrogen dioxide reacts together i.e. \[\text{NO + N}{{\text{O}}_{2}}\to \text{ }{{\text{N}}_{2}}{{\text{O}}_{3}}\].
-Whereas when two molecules of nitrogen oxide dimerise with each other they form nitrogen tetroxide i.e. \[2\text{N}{{\text{O}}_{2}}\ \rightleftharpoons \text{ }{{\text{N}}_{2}}{{\text{O}}_{4}}\]
-Moreover, the oxidation state of nitrogen in nitrogen trioxide, nitrogen tetroxide and nitrogen penta-oxide is +2, +3 and +5.
-So, only nitrogen trioxide and nitrogen tetroxide will have a N - N bond.
Note: Nitrogen can form maximum of 3 covalent bond and 1 co-ordinate bond because according to the electronic configuration it has ability to form 3 bonds easily but in some cases, it can also share some a pair electron i.e. lone pair which forms coordination bond.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




