A solid has a BCC. Structure. If the distance of the closest approach between the two atoms is\[\text{1}\text{.73 Angstrom}\] . The edge length of the cell is (approx.):
A) \[\text{200 pm}\]
B)$\frac{\sqrt{3}}{\sqrt{2}}\text{pm}$
C) \[\text{142}\text{.2 pm}\]
D) $\sqrt{\text{2}}\text{pm}$
Answer
615k+ views
Hint: For BCC structure the radius of the atom ‘r’ and the edge length of the cubic structure ‘a’ are related as:
$\text{r = }\frac{\sqrt{3}\text{a}}{4}$. Use this relation to obtain the answer.
Complete answer:
We are provided with the following data:
a) The Solid has a BCC structure
b) The distance between the atoms,
$\text{2r = 1}\text{.73 }\overset{\text{0}}{\mathop{\text{A}}}\,$
c) To find edge length is ‘a’
-We know that in the BCC structure the atoms are present on the corners of the cubic unit cell and one of the atoms is located at the centre of the body. They are arranged in such a way that the atoms which are on the corners and at the centre of the body are touching each other.
The body-centered cubic structure as follows:
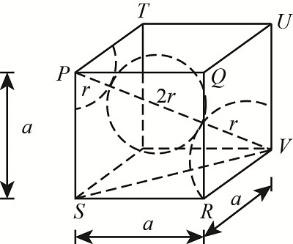
Here ‘a’ represents the length of the edge and ‘r’ is the radius of an atom
The distance between the two atoms $\text{= 2r = 1}\text{.73}\overset{\text{0}}{\mathop{\text{A}}}\,$
Therefore, $\text{= r = }\frac{\text{1}\text{.73}\overset{\text{0}}{\mathop{\text{A}}}\,}{2}=0.865\text{ }\overset{\text{0}}{\mathop{\text{A}}}\,$
For BCC structure, lets us relate the radius ‘r’ with the edge length ‘a’.
The body diagonal PV is composed of a body centered atom and the two atoms at the corners.
Thus, $\text{body diagonal (PV)= r + 2r + r = 4r}$
Let's consider a triangle $\text{ }\!\!\Delta\!\!\text{ SVR}$ ,
Apply Pythagoras theorem. We have,
${{\text{(SV)}}^{\text{2}}}\text{=(SR}{{\text{)}}^{\text{2}}}\text{+(VR}{{\text{)}}^{\text{2}}}$
Where SV is face diagonal. We know that,
$\text{SR=RV=edge length=a}$
Thus,
$\begin{align}
& {{\text{(SV)}}^{\text{2}}}\text{=(a}{{\text{)}}^{\text{2}}}\text{+(a}{{\text{)}}^{\text{2}}} \\
& \text{ = 2}{{\text{a}}^{2}} \\
\end{align}$
Let us apply the Pythagoras theorem in $\text{ }\!\!\Delta\!\!\text{ PSV}$
${{\text{(PV)}}^{\text{2}}}\text{=(PS}{{\text{)}}^{\text{2}}}\text{+(SV}{{\text{)}}^{\text{2}}}$
Substitute the value. We get,
$\begin{align}
& {{\text{(PV)}}^{\text{2}}}\text{=(a}{{\text{)}}^{\text{2}}}\text{+(2a}{{\text{)}}^{\text{2}}} \\
& \text{ = 3}{{\text{a}}^{\text{2}}} \\
\end{align}$
Substitute PV we get,
$\begin{align}
& {{\text{(PV)}}^{\text{2}}}\text{=(a}{{\text{)}}^{\text{2}}}\text{+(2a}{{\text{)}}^{\text{2}}} \\
& \text{ = 3}{{\text{a}}^{\text{2}}} \\
& \text{PV=}\sqrt{\text{3}}\text{a} \\
\end{align}$
Equate the values for PV. We have,
$\text{4r = 3a}$
$\text{r = }\frac{\sqrt{3}\text{a}}{4}$ or $\frac{4}{\sqrt{3}}\text{r = a}$
On substitution values we get,
$\begin{align}
& \text{a=}\frac{\text{4}}{\sqrt{\text{3}}}\text{ }\!\!\times\!\!\text{ (0}\text{.865 }\overset{\text{0}}{\mathop{\text{A}}}\,\text{)} \\
& \text{a=1}\text{.99 }\overset{\text{0}}{\mathop{\text{A}}}\,\text{ }\simeq \text{ 2}\text{.0 }\overset{\text{0}}{\mathop{\text{A}}}\, \\
\end{align}$
The angstrom can be converted into the picometer.
$\text{2}\text{.0 }\overset{\text{0}}{\mathop{\text{A}}}\,=200\text{ pm}$
Hence, (A) is the correct option.
Note:
Do not get confused with the FCC and BCC solid structure. In BCC one atom is at the centre of the body and the other at the corners. However in the FCC structure each face has an atom along with the corners of the body.
$\text{r = }\frac{\sqrt{3}\text{a}}{4}$. Use this relation to obtain the answer.
Complete answer:
We are provided with the following data:
a) The Solid has a BCC structure
b) The distance between the atoms,
$\text{2r = 1}\text{.73 }\overset{\text{0}}{\mathop{\text{A}}}\,$
c) To find edge length is ‘a’
-We know that in the BCC structure the atoms are present on the corners of the cubic unit cell and one of the atoms is located at the centre of the body. They are arranged in such a way that the atoms which are on the corners and at the centre of the body are touching each other.
The body-centered cubic structure as follows:

Here ‘a’ represents the length of the edge and ‘r’ is the radius of an atom
The distance between the two atoms $\text{= 2r = 1}\text{.73}\overset{\text{0}}{\mathop{\text{A}}}\,$
Therefore, $\text{= r = }\frac{\text{1}\text{.73}\overset{\text{0}}{\mathop{\text{A}}}\,}{2}=0.865\text{ }\overset{\text{0}}{\mathop{\text{A}}}\,$
For BCC structure, lets us relate the radius ‘r’ with the edge length ‘a’.
The body diagonal PV is composed of a body centered atom and the two atoms at the corners.
Thus, $\text{body diagonal (PV)= r + 2r + r = 4r}$
Let's consider a triangle $\text{ }\!\!\Delta\!\!\text{ SVR}$ ,
Apply Pythagoras theorem. We have,
${{\text{(SV)}}^{\text{2}}}\text{=(SR}{{\text{)}}^{\text{2}}}\text{+(VR}{{\text{)}}^{\text{2}}}$
Where SV is face diagonal. We know that,
$\text{SR=RV=edge length=a}$
Thus,
$\begin{align}
& {{\text{(SV)}}^{\text{2}}}\text{=(a}{{\text{)}}^{\text{2}}}\text{+(a}{{\text{)}}^{\text{2}}} \\
& \text{ = 2}{{\text{a}}^{2}} \\
\end{align}$
Let us apply the Pythagoras theorem in $\text{ }\!\!\Delta\!\!\text{ PSV}$
${{\text{(PV)}}^{\text{2}}}\text{=(PS}{{\text{)}}^{\text{2}}}\text{+(SV}{{\text{)}}^{\text{2}}}$
Substitute the value. We get,
$\begin{align}
& {{\text{(PV)}}^{\text{2}}}\text{=(a}{{\text{)}}^{\text{2}}}\text{+(2a}{{\text{)}}^{\text{2}}} \\
& \text{ = 3}{{\text{a}}^{\text{2}}} \\
\end{align}$
Substitute PV we get,
$\begin{align}
& {{\text{(PV)}}^{\text{2}}}\text{=(a}{{\text{)}}^{\text{2}}}\text{+(2a}{{\text{)}}^{\text{2}}} \\
& \text{ = 3}{{\text{a}}^{\text{2}}} \\
& \text{PV=}\sqrt{\text{3}}\text{a} \\
\end{align}$
Equate the values for PV. We have,
$\text{4r = 3a}$
$\text{r = }\frac{\sqrt{3}\text{a}}{4}$ or $\frac{4}{\sqrt{3}}\text{r = a}$
On substitution values we get,
$\begin{align}
& \text{a=}\frac{\text{4}}{\sqrt{\text{3}}}\text{ }\!\!\times\!\!\text{ (0}\text{.865 }\overset{\text{0}}{\mathop{\text{A}}}\,\text{)} \\
& \text{a=1}\text{.99 }\overset{\text{0}}{\mathop{\text{A}}}\,\text{ }\simeq \text{ 2}\text{.0 }\overset{\text{0}}{\mathop{\text{A}}}\, \\
\end{align}$
The angstrom can be converted into the picometer.
$\text{2}\text{.0 }\overset{\text{0}}{\mathop{\text{A}}}\,=200\text{ pm}$
Hence, (A) is the correct option.
Note:
Do not get confused with the FCC and BCC solid structure. In BCC one atom is at the centre of the body and the other at the corners. However in the FCC structure each face has an atom along with the corners of the body.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE




