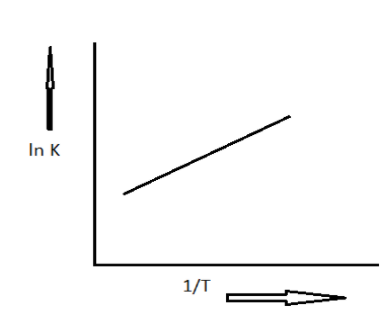
A schematic plot of \[\ln {K_{eq}}\] versus inverse of temperature for a reaction is shown below:
The reaction must be-
A.Exothermic
B.Endothermic
C.One with negligible enthalpy change
D.Highly spontaneous at ordinary temperature

Answer
519k+ views
Hint: The graph describes the equilibrium constant and temperature. The equation can be used to find out the slope. The slope value gives the enthalpy value. From the value of enthalpy, the reaction can be known. The negative enthalpy means releasing of energy which is an exothermic reaction.
Complete answer:
Given graph is drawn between the equilibrium constant and inverse of temperature with a positive slope. The equation can be written in the form of \[y = mx + c\]. Here y is y coordinate which is \[\ln K\] and m is slope which is \[ - \dfrac{{\Delta H}}{R}\] and the X coordinate is inverse of temperature which can be written as \[\dfrac{1}{T}\] and ac is the constant.
Thus, the equation will be
\[\ln K = - \dfrac{{\Delta H}}{{RT}} + C\]
Here the slope is \[ - \dfrac{{\Delta H}}{R}\] , in the given plot the slope is positive.
Thus, this slope must be greater than zero.
\[ - \dfrac{{\Delta H}}{R} > 0\]
By removing the negative sign, we will get
\[\dfrac{{\Delta H}}{R} < 0\]
By simplifying the above relation, we will get
\[\Delta H < 0\]
Thus, the above equation represents the enthalpy is negative. The negative enthalpy favours the exothermic reaction.
Thus, the reaction must be exothermic.
Option A is the correct one.
Note:
Enthalpy is related to heat quantity. The negative enthalpy means the releasing of energy takes place which favours the exothermic reaction, whereas the positive enthalpy means the absorption of energy takes place favours the endothermic reaction.
Complete answer:
Given graph is drawn between the equilibrium constant and inverse of temperature with a positive slope. The equation can be written in the form of \[y = mx + c\]. Here y is y coordinate which is \[\ln K\] and m is slope which is \[ - \dfrac{{\Delta H}}{R}\] and the X coordinate is inverse of temperature which can be written as \[\dfrac{1}{T}\] and ac is the constant.
Thus, the equation will be
\[\ln K = - \dfrac{{\Delta H}}{{RT}} + C\]
Here the slope is \[ - \dfrac{{\Delta H}}{R}\] , in the given plot the slope is positive.
Thus, this slope must be greater than zero.
\[ - \dfrac{{\Delta H}}{R} > 0\]
By removing the negative sign, we will get
\[\dfrac{{\Delta H}}{R} < 0\]
By simplifying the above relation, we will get
\[\Delta H < 0\]
Thus, the above equation represents the enthalpy is negative. The negative enthalpy favours the exothermic reaction.
Thus, the reaction must be exothermic.
Option A is the correct one.
Note:
Enthalpy is related to heat quantity. The negative enthalpy means the releasing of energy takes place which favours the exothermic reaction, whereas the positive enthalpy means the absorption of energy takes place favours the endothermic reaction.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

Difference between unicellular and multicellular o class 11 biology CBSE

Draw a diagram showing the external features of fish class 11 biology CBSE




