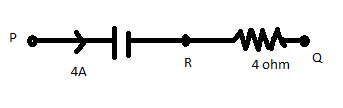
A cell has terminal voltage $2V$ in open circuit and internal resistance of the given cell is $2\Omega $. If $4A$ of current is flowing between points $P$ and $Q$ in the circuit and then the potential difference between $P$ and $Q$ is
A. $22V$
B. $24V$
C. $30V$
D. $26V$

Answer
599.4k+ views
Hint:
Here we have to find the potential difference between the batteries in each terminal and compare them to find the potential difference between $P$ and $Q$.
First let us see what potential difference is-
The potential difference is the difference between two points in a circuit in the amount of energy that the charge carriers have.
Terminal potential difference- Terminal potential difference is usually found in cells or batteries.
The potential difference in a closed circuit between the two poles of a cell is called the cell’s terminal potential difference. When we measure the battery’s emf as we evaluate the potential difference between the terminals of a battery that is not a full circuit. This is the overall amount of work the battery can do to push charge from one terminal through the circuit to the other terminal per coulomb of charge.
Terminal voltage is mathematically represented by-
$V = emf - Ir$
where $emf$ is the electromotive force,
$I$ is the current flowing, and
$r$ is the resistance.
Complete step by step solution-
Given,
A cell has terminal voltage $2V$ in open circuit and internal resistance of the given cell is $2\Omega $
$4A$ of current is flowing between points $P$ and $Q$ in the circuit
$
E = 2V \\
I = 4A \\
r = 4\Omega \\
$
The potential difference between $P$ and $Q$ =?
Case 1
Terminal potential difference
$ = {V_P} - {V_R} = E + Ir = 2 + (4 \times 2) = 10V$
...... (1)
Case 2
Terminal potential difference
$ = {V_R} - {V_Q} = Ir = 4 \times 4 = 16V$
...... (2)
From equation (1) and (2), we get-
$
{V_P} - {V_R} + {V_R} - {V_Q} \\
= 10 + 16 \\
{V_P} - {V_Q} = 26V \\
$
Hence, the potential difference between $P$ and $Q$ is $26V$
Note:
Here as we can see from case (2) we have not added any emf with the second equation. It is because there is no induced emf in the terminal between $R$ and $Q$.
Here we have to find the potential difference between the batteries in each terminal and compare them to find the potential difference between $P$ and $Q$.
First let us see what potential difference is-
The potential difference is the difference between two points in a circuit in the amount of energy that the charge carriers have.
Terminal potential difference- Terminal potential difference is usually found in cells or batteries.
The potential difference in a closed circuit between the two poles of a cell is called the cell’s terminal potential difference. When we measure the battery’s emf as we evaluate the potential difference between the terminals of a battery that is not a full circuit. This is the overall amount of work the battery can do to push charge from one terminal through the circuit to the other terminal per coulomb of charge.
Terminal voltage is mathematically represented by-
$V = emf - Ir$
where $emf$ is the electromotive force,
$I$ is the current flowing, and
$r$ is the resistance.
Complete step by step solution-
Given,
A cell has terminal voltage $2V$ in open circuit and internal resistance of the given cell is $2\Omega $
$4A$ of current is flowing between points $P$ and $Q$ in the circuit
$
E = 2V \\
I = 4A \\
r = 4\Omega \\
$
The potential difference between $P$ and $Q$ =?
Case 1
Terminal potential difference
$ = {V_P} - {V_R} = E + Ir = 2 + (4 \times 2) = 10V$
...... (1)
Case 2
Terminal potential difference
$ = {V_R} - {V_Q} = Ir = 4 \times 4 = 16V$
...... (2)
From equation (1) and (2), we get-
$
{V_P} - {V_R} + {V_R} - {V_Q} \\
= 10 + 16 \\
{V_P} - {V_Q} = 26V \\
$
Hence, the potential difference between $P$ and $Q$ is $26V$
Note:
Here as we can see from case (2) we have not added any emf with the second equation. It is because there is no induced emf in the terminal between $R$ and $Q$.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 Computer Science: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

In cricket, what is the term for a bowler taking five wickets in an innings?

Who Won 36 Oscar Awards? Record Holder Revealed

What is the median of the first 10 natural numbers class 10 maths CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is deficiency disease class 10 biology CBSE




