
A capillary tube of radius ‘r’ is lowered into a liquid of surface tension, T and density, \[\rho \]. The work done by surface tension when the angle of contact is \[{{0}^{0}}\] -
\[\begin{align}
& A)\text{ }\dfrac{\pi {{T}^{2}}}{\rho g} \\
& B)\text{ }\dfrac{4\pi {{T}^{2}}}{\rho g} \\
& C)\text{ }\dfrac{{{T}^{2}}}{\rho g} \\
& D)\text{ }\dfrac{2{{T}^{2}}}{\rho g} \\
\end{align}\]
Answer
591.9k+ views
Hint: We need to find the height of the capillary tube that is immersed in the liquid. From the height given we can calculate the work done by using proper relations. The height can be found using the equation for the surface tension with density term.
Complete answer:
Surface tension is the force acting on the liquid at the place of contact with another medium per unit length. It is also defined as the surface energy or the work done by the liquid per unit area of the liquid. The point of contact plays an important role in the surface energy. The angle of contact determines whether the two media is attracted to each like water on a glass or repelled like mercury on glass.
Let us see how we can find the work done by the liquid in the capillary tube from the given data.
\[\begin{align}
& \text{Surface tension, T}=\dfrac{F}{2\pi r\cos \theta } \\
& \Rightarrow \text{ }F=2\pi Tr\cos \theta \\
\end{align}\]
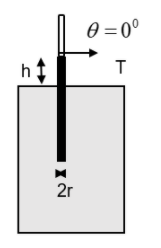
From the figure we can understand that
\[\begin{align}
& \theta ={{0}^{0}} \\
& \Rightarrow \text{ }\cos \theta =1 \\
& \therefore \text{ }F=2\pi Tr \\
\end{align}\]
Now, we can find the height of liquid in the capillary tube by equating the force due to air pressure and the surface tension as –
\[\begin{align}
& F=h\rho g\times \pi {{r}^{2}} \\
& \text{and,} \\
& F=2\pi rT \\
& \Rightarrow \text{ }h\rho g\times \pi {{r}^{2}}=2\pi rT \\
& \Rightarrow \text{ }\rho \pi {{r}^{2}}hg=2\pi rT \\
& \Rightarrow \text{ }h=\dfrac{2T}{\rho rg} \\
\end{align}\]
Now, we can find the work done by the liquid as –
\[\begin{align}
& \text{Work done = Surface tension}\times \operatorname{l}\text{length of contact}\times \text{height of liquid column} \\
& \Rightarrow \text{ }W=T\times 2\pi r\times \dfrac{2T}{\rho rg} \\
& \Rightarrow \text{ }W=\dfrac{4\pi {{T}^{2}}}{\rho g} \\
\end{align}\]
So, we have the work done by the liquid to reach at height ‘h’ as –
\[W=\dfrac{4\pi {{T}^{2}}}{\rho g}\]
The correct answer is given by option B.
Note:
We can find the work done by integrating the surface tension over the area of contact, i.e., in this case the area of the circular face of the capillary tube. We had to find the height in this problem as the options involved didn’t have the height in the equation of the work done.
The work done by the liquid in rising in a capillary tube is the work done against the air pressure.
Complete answer:
Surface tension is the force acting on the liquid at the place of contact with another medium per unit length. It is also defined as the surface energy or the work done by the liquid per unit area of the liquid. The point of contact plays an important role in the surface energy. The angle of contact determines whether the two media is attracted to each like water on a glass or repelled like mercury on glass.
Let us see how we can find the work done by the liquid in the capillary tube from the given data.
\[\begin{align}
& \text{Surface tension, T}=\dfrac{F}{2\pi r\cos \theta } \\
& \Rightarrow \text{ }F=2\pi Tr\cos \theta \\
\end{align}\]

From the figure we can understand that
\[\begin{align}
& \theta ={{0}^{0}} \\
& \Rightarrow \text{ }\cos \theta =1 \\
& \therefore \text{ }F=2\pi Tr \\
\end{align}\]
Now, we can find the height of liquid in the capillary tube by equating the force due to air pressure and the surface tension as –
\[\begin{align}
& F=h\rho g\times \pi {{r}^{2}} \\
& \text{and,} \\
& F=2\pi rT \\
& \Rightarrow \text{ }h\rho g\times \pi {{r}^{2}}=2\pi rT \\
& \Rightarrow \text{ }\rho \pi {{r}^{2}}hg=2\pi rT \\
& \Rightarrow \text{ }h=\dfrac{2T}{\rho rg} \\
\end{align}\]
Now, we can find the work done by the liquid as –
\[\begin{align}
& \text{Work done = Surface tension}\times \operatorname{l}\text{length of contact}\times \text{height of liquid column} \\
& \Rightarrow \text{ }W=T\times 2\pi r\times \dfrac{2T}{\rho rg} \\
& \Rightarrow \text{ }W=\dfrac{4\pi {{T}^{2}}}{\rho g} \\
\end{align}\]
So, we have the work done by the liquid to reach at height ‘h’ as –
\[W=\dfrac{4\pi {{T}^{2}}}{\rho g}\]
The correct answer is given by option B.
Note:
We can find the work done by integrating the surface tension over the area of contact, i.e., in this case the area of the circular face of the capillary tube. We had to find the height in this problem as the options involved didn’t have the height in the equation of the work done.
The work done by the liquid in rising in a capillary tube is the work done against the air pressure.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Actinoid contraction is more than lanthanoid contraction class 11 chemistry CBSE

The transition element that has lowest enthalpy of class 11 chemistry CBSE

Can anyone list 10 advantages and disadvantages of friction

State the laws of reflection of light




