2,6 - Dimethylheptane on monochlorination produces..................derivatives.
A) 5
B) 6
C) 3
D) 4
Answer
512.7k+ views
Hint: Monochlorination is the addition of chlorine at the terminal methyl groups. The chlorine atom replaces the hydrogen from the methyl group and gets attached to the carbon of the methyl group. Monochlorination means only single chlorine is being added, although multiple chlorine atoms can bind with the molecule based on the availability of methyl groups.
Complete answer:
2,6 - Dimethylheptane is a disubstituted alkane. Two methyl groups are attached to the second and sixth carbon atoms on the chain. The structure of the 2,6 - Dimethylheptane can be drawn as-
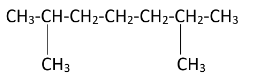
As the diagram suggests, there are four free terminal methyl groups. Monochlorination can occur on all of them. The products formed after mono chlorination will be -
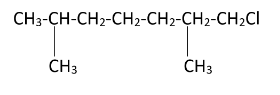
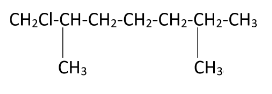
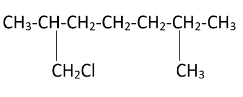
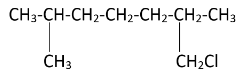
Hence, we can see that there are four possible monochlorination products.
So the correct option is D) 4.
Note:
The products formed can be named as – [7-chloro, 2,6 dimethylheptane], [1-chloro, 2,6 dimethylheptane], [2-chloromethyl, 6-methyl heptane], [6-chloromethyl, 2-methyl heptane]. Simultaneous chlorination can also occur at all the methyl groups present in the molecule resulting in tetrachloro substitution.
Complete answer:
2,6 - Dimethylheptane is a disubstituted alkane. Two methyl groups are attached to the second and sixth carbon atoms on the chain. The structure of the 2,6 - Dimethylheptane can be drawn as-
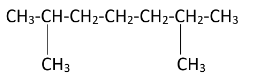
As the diagram suggests, there are four free terminal methyl groups. Monochlorination can occur on all of them. The products formed after mono chlorination will be -
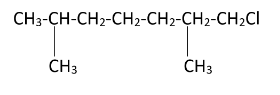
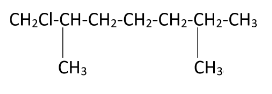
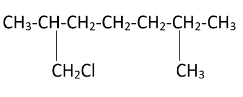
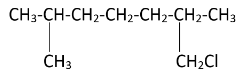
Hence, we can see that there are four possible monochlorination products.
So the correct option is D) 4.
Note:
The products formed can be named as – [7-chloro, 2,6 dimethylheptane], [1-chloro, 2,6 dimethylheptane], [2-chloromethyl, 6-methyl heptane], [6-chloromethyl, 2-methyl heptane]. Simultaneous chlorination can also occur at all the methyl groups present in the molecule resulting in tetrachloro substitution.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




