1-phenyl-2-chloropropane on treating with alc. KOH gives mainly.
A. 1-phenyl propene
B. 2-phenyl propene
C. 1-phenylpropan-2-ol
D. 1-phenyl-propan-1-ol
Answer
600.3k+ views
Hint: In the given reaction, dehydrohalogenation is taking place which means the chloride ion is removed which further acts as a nucleophile and attacks the metal cation to form salt. In this reaction the alcoholic potassium hydroxide acts as a base. The main product formed will be an unsaturated hydrocarbon compound.
Complete step by step answer:
In the given reaction, a dehydrohalogenation reaction is taking place. In this reaction, the alcoholic KOH is acting as a base where potassium carries a positive charge and the hydroxyl group carries the negative charge. The hydroxyl anion will abstract the hydrogen present at the alpha position of 1-phenyl-2-chloropropane to eliminate water. After the removal of the hydrogen the charge will shift to form a double bond and chlorine will be removed as chloride anion. The chloride anion will further act as a nucleophile and will attack the positive charged potassium to form potassium chloride. The resulting main product which will be formed is 1-phenyl propene
The reaction between 1-phenyl-2-chloropropane with alc. KOH is shown below.
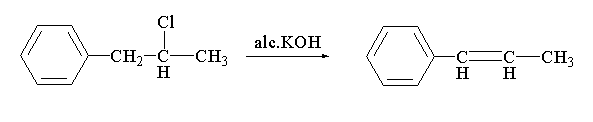
In this reaction, 1-phenyl-2-chloropropane reacts with alcoholic potassium hydroxide to form 1-phenyl propene.
Therefore, the correct option is A.
Note: The reaction is a type of elimination reaction, as a saturated compound is converted to an unsaturated compound. If in place of alcoholic potassium hydroxide, aqueous potassium hydroxide solution was used, the resulting product which will be formed is alcohol.
Complete step by step answer:
In the given reaction, a dehydrohalogenation reaction is taking place. In this reaction, the alcoholic KOH is acting as a base where potassium carries a positive charge and the hydroxyl group carries the negative charge. The hydroxyl anion will abstract the hydrogen present at the alpha position of 1-phenyl-2-chloropropane to eliminate water. After the removal of the hydrogen the charge will shift to form a double bond and chlorine will be removed as chloride anion. The chloride anion will further act as a nucleophile and will attack the positive charged potassium to form potassium chloride. The resulting main product which will be formed is 1-phenyl propene
The reaction between 1-phenyl-2-chloropropane with alc. KOH is shown below.

In this reaction, 1-phenyl-2-chloropropane reacts with alcoholic potassium hydroxide to form 1-phenyl propene.
Therefore, the correct option is A.
Note: The reaction is a type of elimination reaction, as a saturated compound is converted to an unsaturated compound. If in place of alcoholic potassium hydroxide, aqueous potassium hydroxide solution was used, the resulting product which will be formed is alcohol.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




