Write two examples of metallic solids.
Answer
596.7k+ views
Hint: Solid is one of the fundamental states of matter. The molecules in solid are closely packed together and contain the least amount of kinetic energy. They are rigid and generally harder than liquid and gas. Metallic solids generally possess cubic crystal structures.
Complete Step by step answer: metallic solids are solid composed of metal atoms that are held together by metallic bonds. In these solids positive ions are surrounded by mobile free electrons and are evenly spread all over the crystal. The electrons in the metallic solids are delocalized. They are not just held between a couple of atoms in a sigma bond. Rather there is a sea of electrons everywhere.
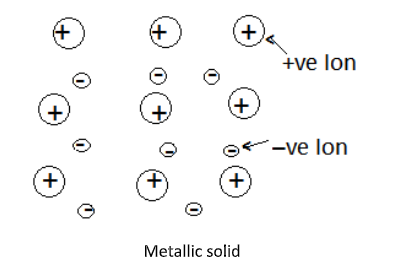
Each metal atom donates one or more electrons to the group of mobile electrons which increases the electrical and thermal conductivity of the metallic elements.
Application of electric fields makes these electrons flow through the linkage of positive ions, hence they are good conductors of electricity. Whereas application of heat to one portion of metal makes the thermal energy spread uniformly throughout by free electrons.
Metals are malleable which means they can be pounded into sheets. They are also ductile, which means they can be pulled into wires. The main idea is that unlike covalent solids, they are stiff or brittle. That is because the cations in the sea of electrons can slide around without needing to break any very strong bond.
Examples of metallic solids – copper, gold, zinc etc.
Note: We generally think metallic solids as pure metals, but they can also be the combinations of metals that are alloys like bronze which is a mixture of copper and tin. Metallic solids because of the free electrons, are good conductors of electricity.
Complete Step by step answer: metallic solids are solid composed of metal atoms that are held together by metallic bonds. In these solids positive ions are surrounded by mobile free electrons and are evenly spread all over the crystal. The electrons in the metallic solids are delocalized. They are not just held between a couple of atoms in a sigma bond. Rather there is a sea of electrons everywhere.

Each metal atom donates one or more electrons to the group of mobile electrons which increases the electrical and thermal conductivity of the metallic elements.
Application of electric fields makes these electrons flow through the linkage of positive ions, hence they are good conductors of electricity. Whereas application of heat to one portion of metal makes the thermal energy spread uniformly throughout by free electrons.
Metals are malleable which means they can be pounded into sheets. They are also ductile, which means they can be pulled into wires. The main idea is that unlike covalent solids, they are stiff or brittle. That is because the cations in the sea of electrons can slide around without needing to break any very strong bond.
Examples of metallic solids – copper, gold, zinc etc.
Note: We generally think metallic solids as pure metals, but they can also be the combinations of metals that are alloys like bronze which is a mixture of copper and tin. Metallic solids because of the free electrons, are good conductors of electricity.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which Indian city is known as the "City of Victory"?

Which instrument is used to measure the Blood Pressure?

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

Distinguish between the reserved forests and protected class 10 biology CBSE

Who hosted the Congress of Vienna in 1815 A Duke Metternich class 10 social science CBSE

Fill in the blank using the plural form of the noun class 10 english CBSE




