Write the Rutherford -Soddy law of radioactive decay and draw decay curve.
Answer
612.9k+ views
Hint:The Rutherford -Soddy law tells us about the radiation and decay of elements.
Formula used: \[N={{N}_{0}}{{e}^{-\lambda t}}\]
Complete Answer:
1) The radioactivity is based on counting of disintegration per second. The activity depends on the number of decays per second.
2) The law is the exponential decay law which states that a fixed fraction of elements will decay per unit time. Let us take the example, the half part of thorium decays and the remaining in the next four days and so on.
The derivation of the formula: -
\[\begin{align}
& -\dfrac{dN}{dt}\alpha N \\
& \Rightarrow -\dfrac{dN}{dt}=kN \\
& \Rightarrow \int\limits_{{{N}_{0}}}^{N}{\dfrac{dN}{N}}=\lambda \int\limits_{0}^{t}{dt} \\
& \Rightarrow N={{N}_{0}}{{e}^{-\lambda t}} \\
\end{align}\]
where, N= size of population of radioactive element , N₀= size of initial population of radioactive element, 𝝀 = decay constant.
This shows that the population decays exponentially at a rate that depends on the decay constant.
The time required for half of the population to decay is called the half-life which is given by
\[{{T}_{{}^{1}/{}_{2}}}=\dfrac{0.693}{\lambda }\]
The decay curve is shown in the figure below:
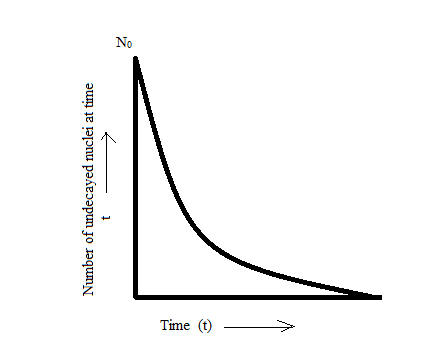
Note: Radioactivity property is exhibited by certain types of matter of emitting energy simultaneously. Half-life is the time required for an element to reduce to half of its initial value.
Formula used: \[N={{N}_{0}}{{e}^{-\lambda t}}\]
Complete Answer:
1) The radioactivity is based on counting of disintegration per second. The activity depends on the number of decays per second.
2) The law is the exponential decay law which states that a fixed fraction of elements will decay per unit time. Let us take the example, the half part of thorium decays and the remaining in the next four days and so on.
The derivation of the formula: -
\[\begin{align}
& -\dfrac{dN}{dt}\alpha N \\
& \Rightarrow -\dfrac{dN}{dt}=kN \\
& \Rightarrow \int\limits_{{{N}_{0}}}^{N}{\dfrac{dN}{N}}=\lambda \int\limits_{0}^{t}{dt} \\
& \Rightarrow N={{N}_{0}}{{e}^{-\lambda t}} \\
\end{align}\]
where, N= size of population of radioactive element , N₀= size of initial population of radioactive element, 𝝀 = decay constant.
This shows that the population decays exponentially at a rate that depends on the decay constant.
The time required for half of the population to decay is called the half-life which is given by
\[{{T}_{{}^{1}/{}_{2}}}=\dfrac{0.693}{\lambda }\]
The decay curve is shown in the figure below:

Note: Radioactivity property is exhibited by certain types of matter of emitting energy simultaneously. Half-life is the time required for an element to reduce to half of its initial value.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




