While separating a mixture of ortho and para- nitrophenol by steam distillation,name the isomer which will be steam volatile? Give reasons.
Answer
586.2k+ views
Hint: We should try checking the hydrogen bonding between the two compounds and that might give us an idea about their boiling point.So,in order to distinguish both of the isomers,we should try check the positions of the functional groups here.
Complete answer:
Ortho-nitrophenol shows intramolecular hydrogen bonding, which means bonding is present between it’s own molecule,between the phenol and nitro groups. Owing to this, when the given compound is subjected to steam distillation,the compound easily vaporizes out ,thus having a low boiling point and thus will be steam volatile.
Let’s try to understand by the diagram of the compound:
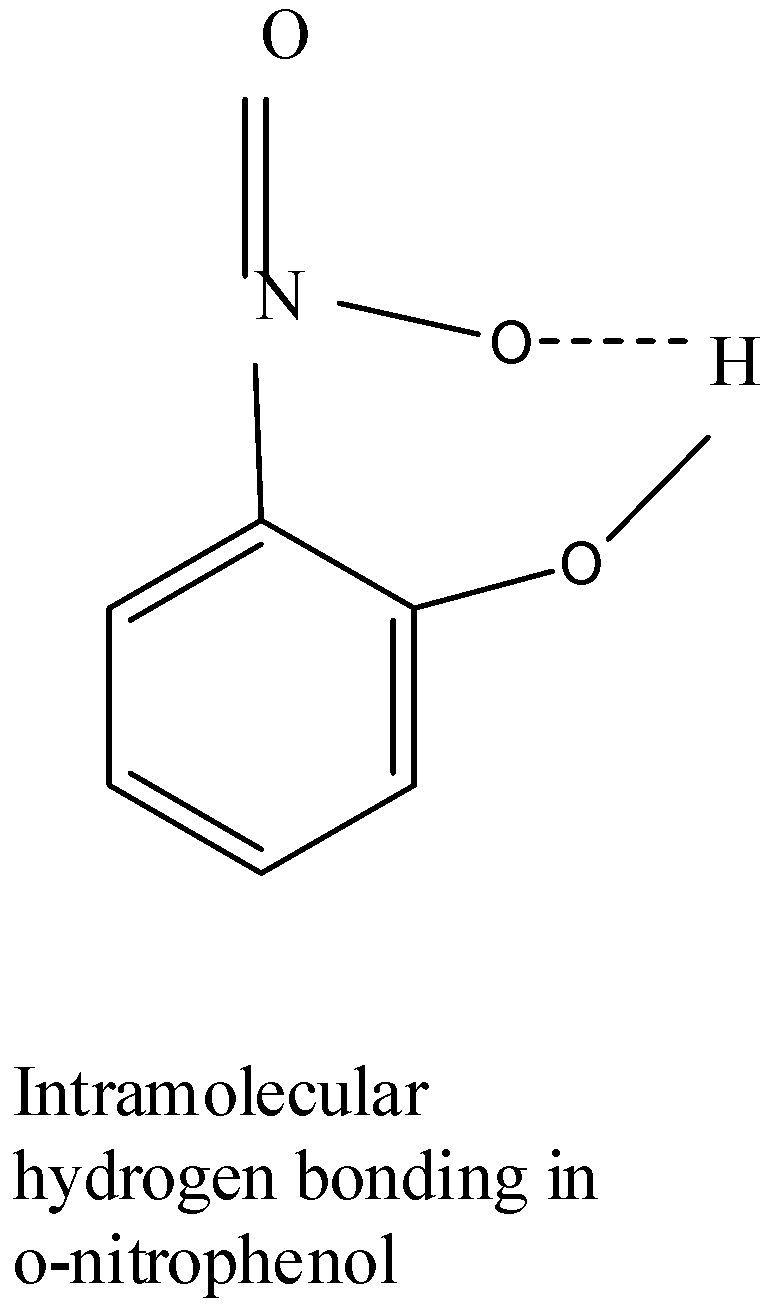
So, from the structure it is clear that it shows intramolecular hydrogen bonding.
The boiling point of ortho-nitrophenol is \[\text{21}{{\text{6}}^{\text{o}}}\text{C}\]..
Now let us consider the compound para-nitrophenol
This structure ,shows intermolecular hydrogen bonding,which means that it can show bonding with other molecules, and thus it has a strong bond which when tried to separate by steam distillation, shows less volatility,as this type of bonding leads to molecular association.Therefore, this compound does not easily evaporates out, due to which it has higher boiling point and does not separate during the process.
Let us try to understand it’s structure:
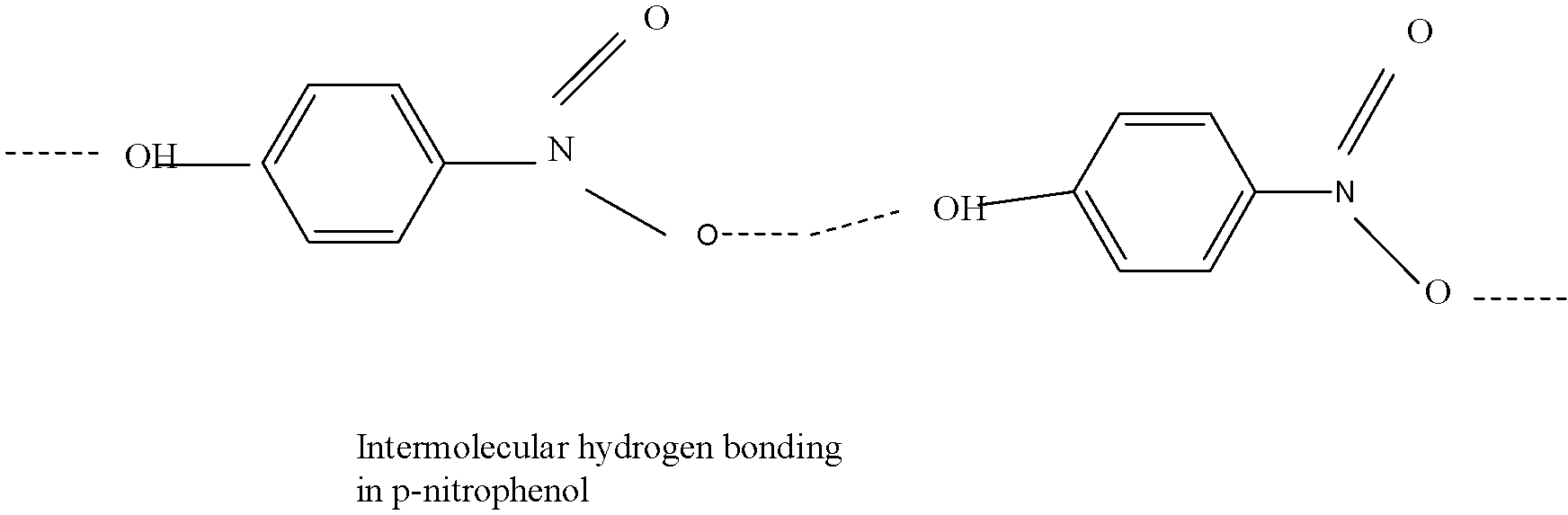
Now looking at the structure,it is prevalent that it shows bonding between its other molecules, and thus also shows a higher boiling point.
The boiling point of para nitrophenol is \[\text{27}{{\text{9}}^{\text{o}}}\text{C}\]
Thus, we can infer that the ortho-nitrophenol is steam volatile then the para-nitrophenol owing to the above stated reasons.
Note:
We should remember that the steam volatile nature talks about the boiling point,which is checked by the type of bonding both the compounds show.The position of the isomers is of utmost importance.
Complete answer:
Ortho-nitrophenol shows intramolecular hydrogen bonding, which means bonding is present between it’s own molecule,between the phenol and nitro groups. Owing to this, when the given compound is subjected to steam distillation,the compound easily vaporizes out ,thus having a low boiling point and thus will be steam volatile.
Let’s try to understand by the diagram of the compound:

So, from the structure it is clear that it shows intramolecular hydrogen bonding.
The boiling point of ortho-nitrophenol is \[\text{21}{{\text{6}}^{\text{o}}}\text{C}\]..
Now let us consider the compound para-nitrophenol
This structure ,shows intermolecular hydrogen bonding,which means that it can show bonding with other molecules, and thus it has a strong bond which when tried to separate by steam distillation, shows less volatility,as this type of bonding leads to molecular association.Therefore, this compound does not easily evaporates out, due to which it has higher boiling point and does not separate during the process.
Let us try to understand it’s structure:

Now looking at the structure,it is prevalent that it shows bonding between its other molecules, and thus also shows a higher boiling point.
The boiling point of para nitrophenol is \[\text{27}{{\text{9}}^{\text{o}}}\text{C}\]
Thus, we can infer that the ortho-nitrophenol is steam volatile then the para-nitrophenol owing to the above stated reasons.
Note:
We should remember that the steam volatile nature talks about the boiling point,which is checked by the type of bonding both the compounds show.The position of the isomers is of utmost importance.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




