Which pair of statements about diamond and graphite is correct?
A.Diamond and graphite are both pure carbons. They are both macromolecules.
B.Diamond and graphite can both be used as electrodes. Graphite is also used as a lubricant.
C.Diamond has covalent bonds. Graphite has ionic bonds.
D.Diamond is hard with a high melting point. Graphite is soft with a low melting point.
Answer
610.5k+ views
Hint: Allotropy is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Diamond, Graphite and Fullerene are popular allotropes of Carbon. Diamond is the hardest substance in the world. Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure.
Complete step by step answer:
Graphite is the most stable allotrope of carbon. Under normal conditions, the diamond is metastable, meaning that it converts back to graphite when the process is initiated with sufficient energy. All allotrope of carbon turns into graphite at higher temperatures. Diamond also turns into graphite at a temperature between $1800 - {2000^ \circ }C$ .
Diamond cannot conduct electricity. On the other hand, graphite can conduct electricity. Therefore, the diamond cannot be used as an electrode. Graphite and carbon both are macromolecules of carbon only with a different arrangement of carbon molecules. In graphite and diamond both have covalent bonds. Both graphite and diamond have the boiling point at \[{3600^\circ }C\] . The structures are shown below,
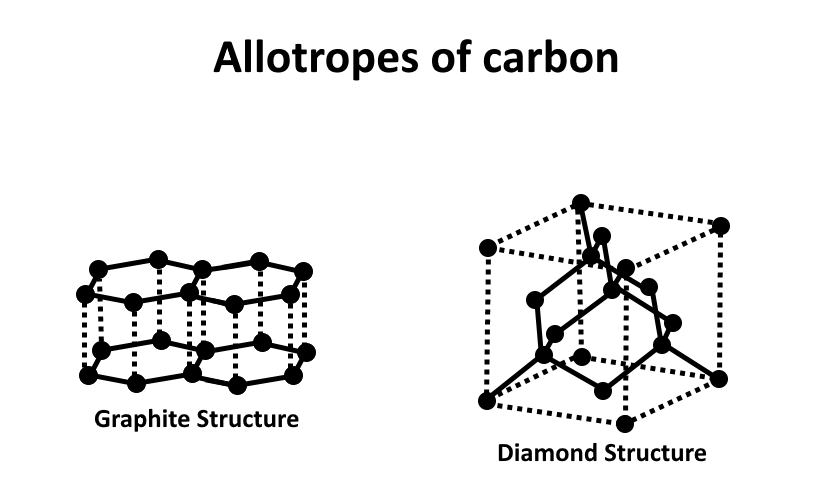
So, the correct option is, A.
Note: Diamond is harder than graphite because, in diamond, each carbon atom forms 4 covalent bonds in a tetrahedral structure. The sheets of carbon become bonded by weaker intermolecular forces. It is because of these weak intermolecular forces that the layers of graphite can slide over each other, making the overall substance a lot weaker than diamond.
Complete step by step answer:
Graphite is the most stable allotrope of carbon. Under normal conditions, the diamond is metastable, meaning that it converts back to graphite when the process is initiated with sufficient energy. All allotrope of carbon turns into graphite at higher temperatures. Diamond also turns into graphite at a temperature between $1800 - {2000^ \circ }C$ .
Diamond cannot conduct electricity. On the other hand, graphite can conduct electricity. Therefore, the diamond cannot be used as an electrode. Graphite and carbon both are macromolecules of carbon only with a different arrangement of carbon molecules. In graphite and diamond both have covalent bonds. Both graphite and diamond have the boiling point at \[{3600^\circ }C\] . The structures are shown below,

So, the correct option is, A.
Note: Diamond is harder than graphite because, in diamond, each carbon atom forms 4 covalent bonds in a tetrahedral structure. The sheets of carbon become bonded by weaker intermolecular forces. It is because of these weak intermolecular forces that the layers of graphite can slide over each other, making the overall substance a lot weaker than diamond.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




