Which one of the following statements is correct?
A.All amino acids except lysine are optically active.
B.All amino acids are optically active.
C.All amino acids except glycine are optically active.
D.All amino acids except glutamic acids are optically active.
Answer
576.6k+ views
Hint: Amino acids are building blocks of proteins. Optical activity of amino acids arises due to the presence of asymmetric carbon. Asymmetric carbon or chiral carbon is one which is attached to four different groups.
Complete step by step answer:
All amino acids contain amino groups and carboxyl groups and hydrogen. A central carbon is attached to these three groups. The property of amino acid is determined by the fourth group which is attached to this carbon. Based on this group, amino acids can be acidic, basic or neutral. Optical activity of amino acid also depends on this fourth group.
Presence of chiral carbon makes the molecule optically active. In amino acids, if the four groups present are different, then the amino acid will be optically active.
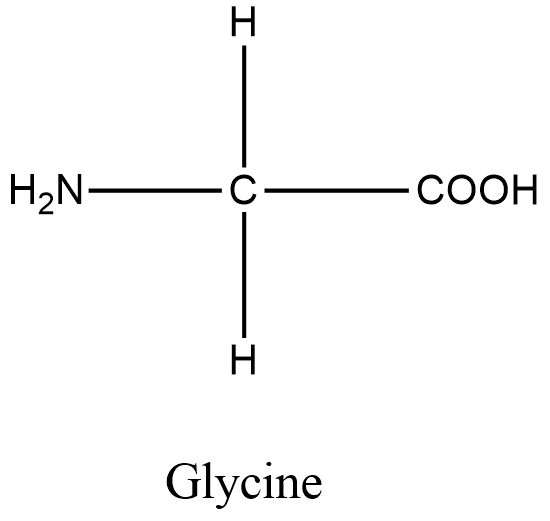
In glycine, the fourth group present is hydrogen. Since there are two hydrogens present, glycine is optically inactive. All other amino acids contain four different groups. Hence all other amino acids are optically active. Therefore the correct statement is, all amino acids except glycine are optically active. Option C is correct.
Structure of glycine is shown in the figure.
Additional information-
The carbon to which both amino and carboxyl groups are attached is called $\alpha - $carbon.
Note:
Lysine and glutamic acid are optically active amino acids. They contain four different groups on $\alpha - $carbon. Asymmetry at the polypeptide chain also makes the amino acids optically active. Optically active amino acids can rotate the plane of polarisation of plane polarised light either to right or left. Accordingly they are classified as D- or L- isomers.
Complete step by step answer:
All amino acids contain amino groups and carboxyl groups and hydrogen. A central carbon is attached to these three groups. The property of amino acid is determined by the fourth group which is attached to this carbon. Based on this group, amino acids can be acidic, basic or neutral. Optical activity of amino acid also depends on this fourth group.
Presence of chiral carbon makes the molecule optically active. In amino acids, if the four groups present are different, then the amino acid will be optically active.
In glycine, the fourth group present is hydrogen. Since there are two hydrogens present, glycine is optically inactive. All other amino acids contain four different groups. Hence all other amino acids are optically active. Therefore the correct statement is, all amino acids except glycine are optically active. Option C is correct.
Structure of glycine is shown in the figure.

Additional information-
The carbon to which both amino and carboxyl groups are attached is called $\alpha - $carbon.
Note:
Lysine and glutamic acid are optically active amino acids. They contain four different groups on $\alpha - $carbon. Asymmetry at the polypeptide chain also makes the amino acids optically active. Optically active amino acids can rotate the plane of polarisation of plane polarised light either to right or left. Accordingly they are classified as D- or L- isomers.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




