Which of the following statements is incorrect?
a.) The frequency of radiation is inversely proportional to its wavelength.
b.) The energy of radiation increases with an increase in frequency.
c.) The energy of radiation decreases with an increase in wavelength.
d.) The frequency of radiation is directly proportional to its wavelength.
Answer
630k+ views
Hint: Wavelength is the distance between identical points in the adjacent cycles of a waveform signal propagated in space or along a wire. Wavelength is inversely related to frequency. The higher the frequency of the signal, the shorter the wavelength.
Complete step by step solution:
Wavelength is inversely proportional to the frequency of the wave. The waves with higher frequencies have shorter wavelengths, and lower frequencies have longer wavelengths.
As we know that, E=hν
Where, E = Energy of the radiation
h = Planck’s constant
${ \mu }$ = frequency of radiation
As we see the energy of the radiation is directly proportional to the frequency of the radiation. Hence, we can say that as the energy of radiation increases, the frequency also increases and vice versa.
As we know, ${ E=h\upsilon =hc\div \lambda }$
Where, E = Energy of the radiation
h = Planck’s constant
${ \mu }$ = frequency of radiation
${\lambda}$ = wavelength of the radiation
Energy is directly proportional to frequency and frequency is inversely proportional to the wavelength of the radiation. So, as the energy decreases, wavelength increases and vice versa as the energy of the radiation is inversely proportional to the wavelength of the radiation.
${ \upsilon =c\div \lambda }$
where, ${ \mu }$ = frequency of radiation
${\lambda}$ = wavelength of the radiation
c =speed of light
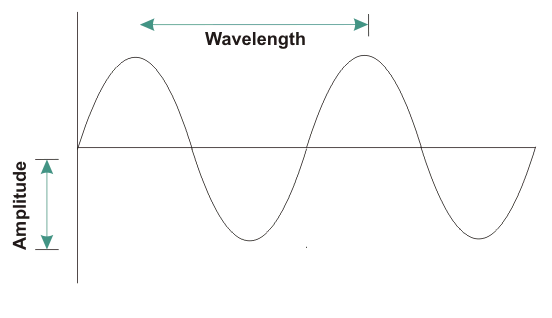
From here, we see that frequency of the radiation is inversely proportional to the wavelength of the radiation. So, the above statement is incorrect.
So, the correct answer is “Option D”.
Note: The possibility to make a mistake is that you may confuse between A and D. But as we know the frequency of the radiation is inversely proportional to the frequency of the radiation, so option D has an incorrect statement.
Complete step by step solution:
Wavelength is inversely proportional to the frequency of the wave. The waves with higher frequencies have shorter wavelengths, and lower frequencies have longer wavelengths.
As we know that, E=hν
Where, E = Energy of the radiation
h = Planck’s constant
${ \mu }$ = frequency of radiation
As we see the energy of the radiation is directly proportional to the frequency of the radiation. Hence, we can say that as the energy of radiation increases, the frequency also increases and vice versa.
As we know, ${ E=h\upsilon =hc\div \lambda }$
Where, E = Energy of the radiation
h = Planck’s constant
${ \mu }$ = frequency of radiation
${\lambda}$ = wavelength of the radiation
Energy is directly proportional to frequency and frequency is inversely proportional to the wavelength of the radiation. So, as the energy decreases, wavelength increases and vice versa as the energy of the radiation is inversely proportional to the wavelength of the radiation.
${ \upsilon =c\div \lambda }$
where, ${ \mu }$ = frequency of radiation
${\lambda}$ = wavelength of the radiation
c =speed of light

From here, we see that frequency of the radiation is inversely proportional to the wavelength of the radiation. So, the above statement is incorrect.
So, the correct answer is “Option D”.
Note: The possibility to make a mistake is that you may confuse between A and D. But as we know the frequency of the radiation is inversely proportional to the frequency of the radiation, so option D has an incorrect statement.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




