Which of the following reactions will best convert nitrobenzene into 2, 4, 6-trichlorobromobenzene?
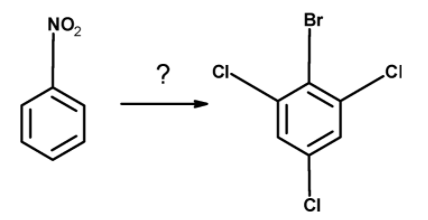
A) $\text{ SnC}{{\text{l}}_{\text{2}}}\text{ / HCl , C}{{\text{l}}_{\text{2}}}-{{\text{H}}_{\text{2}}}\text{O , NaN}{{\text{O}}_{\text{2}}}\text{ / HCl }-{{\text{0}}^{\text{0}}}\text{C , CuBr }$
B) $\text{ C}{{\text{l}}_{\text{2}}}\text{ / AlC}{{\text{l}}_{\text{3}}}\text{ , SnC}{{\text{l}}_{\text{2}}}\text{/HCl , NaN}{{\text{O}}_{\text{2}}}\text{ / HCl }-{{\text{0}}^{\text{0}}}\text{C , CuBr }$
C) $\text{ SnC}{{\text{l}}_{\text{2}}}\text{ / HCl , B}{{\text{r}}_{\text{2}}}-{{\text{H}}_{\text{2}}}\text{O , NaN}{{\text{O}}_{\text{2}}}\text{ / HCl }-{{\text{0}}^{\text{0}}}\text{C , CuCl }$
D) $\text{ SnC}{{\text{l}}_{\text{2}}}\text{ / HCl , NaN}{{\text{O}}_{\text{2}}}\text{ / HCl }-{{\text{0}}^{\text{0}}}\text{C , }{{\text{H}}_{\text{2}}}\text{O , C}{{\text{l}}_{\text{2}}}\text{(aq) , HBr }$

Answer
598.5k+ views
Hint: The aromatic nitro compounds are reduced by the active metals. The nitro group is reduced to $\text{ }-\text{N}{{\text{H}}_{\text{2}}}\text{ }$ groups. The exposure to the excess of halogen results in the 2, 4, 6-tri halo substituted benzene ring. The reaction of the conversion of aromatic primary amines to the diazonium salt $\text{ ArN}_{2}^{+}{{\text{X}}^{-}}\text{ }$is a diazotization reaction. The diazonium group in the diazonium salts is replaced by the groups to get the desired product.
Complete answer:
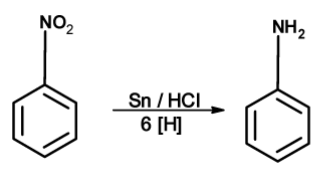
The nitro compounds can be reduced with active metals such as $\text{ Fe }$ , $\text{ Sn }$ , $\text{ Zn }$ , etc. And conc. hydrochloric acid. The mixture of $\text{ SnC}{{\text{l}}_{\text{2}}}\text{ }$ and conc.$\text{ HCl }$ has been used for the reduction of aromatic nitro compounds.
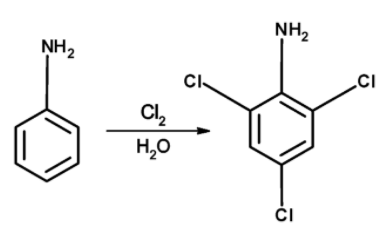
Aniline when reacted with the chlorine molecules, the aniline is converted into 2, 4, 6-trichloroaniline. The reaction of conversion of aniline to the 2, 4, 6-trichloroaniline is as follows,
The diazonium salts have the general formula of $\text{ ArN}_{2}^{+}{{\text{X}}^{-}}\text{ }$ , where $\text{ }{{\text{X}}^{-}}\text{ }$ maybe anion like chloride, bromide, etc. and the group $\text{ N}_{2}^{+\text{ }}$ ($\text{ }-\text{N }\equiv \text{ N}-\text{ }$) is called as the diazonium group. These are obtained when the aromatic primary amines react with nitrous acid.
The diazonium salt is prepared by heating the ice-cold solution of aromatic primary amines in excess of the mineral acid like $\text{ HCl }$ or $\text{ }{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\text{ }$ with the ice-cold solution of sodium nitrite dissolved in water. The temperature is maintained between the $\text{ 273 K }-278\text{ K }$ because most of the diazonium salts decompose at a higher temperature.
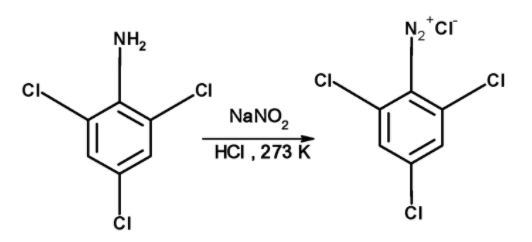
The benzene diazonium chloride of The 2, 4, 6-trichloroaniline is prepared by treating an ice-cold solution of aniline in hydrochloric acid with an ice-cold solution of sodium nitrite at about $\text{ }{{\text{0}}^{\text{0}}}\text{C }$ . The reaction of converting aromatic primary amine to diazonium salt is called diazotization. The 2, 4, 6-trichloroaniline is converted into diazonium salt as follows,
$\text{ NaN}{{\text{O}}_{\text{2}}}\text{ + HCl }\to \text{ NaCl + HONO }$
The diazonium salt of The 2, 4, 6-trichloroaniline when warmed with the cuprous bromide $\text{ CuBr }$ in hydrochloric acid the corresponding bromide is formed. The resultant compound is 2, 4, 6-trichlorobromobenzene. The diazonium salt is converted to the halide group. The reaction is as follows,

Thus, the nitrobenzene is converted into the 2, 4, 6-trichlorobromobenzene by the action of the following reagent : $\text{ SnC}{{\text{l}}_{\text{2}}}\text{ / HCl , C}{{\text{l}}_{\text{2}}}-{{\text{H}}_{\text{2}}}\text{O , NaN}{{\text{O}}_{\text{2}}}\text{ / HCl }-{{\text{0}}^{\text{0}}}\text{C , CuBr }$
Hence, (A) is the correct option.
Note:
Note that the preparation of aryl halide from diazonium salt is a good method and has many advantages. Aryl chlorides and bromides when obtained by direct halogenation of aromatic compounds give a mixture of products that are difficult to separate. However, in the diazonium salt replacement method, a pure single product is formed. This also provides a good synthetic route for the preparation of aryl iodides and fluorides which are not obtained by direct halogenation.
Complete answer:
The nitro compounds can be reduced with active metals such as $\text{ Fe }$ , $\text{ Sn }$ , $\text{ Zn }$ , etc. And conc. hydrochloric acid. The mixture of $\text{ SnC}{{\text{l}}_{\text{2}}}\text{ }$ and conc.$\text{ HCl }$ has been used for the reduction of aromatic nitro compounds.

Aniline when reacted with the chlorine molecules, the aniline is converted into 2, 4, 6-trichloroaniline. The reaction of conversion of aniline to the 2, 4, 6-trichloroaniline is as follows,

The diazonium salts have the general formula of $\text{ ArN}_{2}^{+}{{\text{X}}^{-}}\text{ }$ , where $\text{ }{{\text{X}}^{-}}\text{ }$ maybe anion like chloride, bromide, etc. and the group $\text{ N}_{2}^{+\text{ }}$ ($\text{ }-\text{N }\equiv \text{ N}-\text{ }$) is called as the diazonium group. These are obtained when the aromatic primary amines react with nitrous acid.
The diazonium salt is prepared by heating the ice-cold solution of aromatic primary amines in excess of the mineral acid like $\text{ HCl }$ or $\text{ }{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\text{ }$ with the ice-cold solution of sodium nitrite dissolved in water. The temperature is maintained between the $\text{ 273 K }-278\text{ K }$ because most of the diazonium salts decompose at a higher temperature.
The benzene diazonium chloride of The 2, 4, 6-trichloroaniline is prepared by treating an ice-cold solution of aniline in hydrochloric acid with an ice-cold solution of sodium nitrite at about $\text{ }{{\text{0}}^{\text{0}}}\text{C }$ . The reaction of converting aromatic primary amine to diazonium salt is called diazotization. The 2, 4, 6-trichloroaniline is converted into diazonium salt as follows,
$\text{ NaN}{{\text{O}}_{\text{2}}}\text{ + HCl }\to \text{ NaCl + HONO }$

The diazonium salt of The 2, 4, 6-trichloroaniline when warmed with the cuprous bromide $\text{ CuBr }$ in hydrochloric acid the corresponding bromide is formed. The resultant compound is 2, 4, 6-trichlorobromobenzene. The diazonium salt is converted to the halide group. The reaction is as follows,

Thus, the nitrobenzene is converted into the 2, 4, 6-trichlorobromobenzene by the action of the following reagent : $\text{ SnC}{{\text{l}}_{\text{2}}}\text{ / HCl , C}{{\text{l}}_{\text{2}}}-{{\text{H}}_{\text{2}}}\text{O , NaN}{{\text{O}}_{\text{2}}}\text{ / HCl }-{{\text{0}}^{\text{0}}}\text{C , CuBr }$
Hence, (A) is the correct option.
Note:
Note that the preparation of aryl halide from diazonium salt is a good method and has many advantages. Aryl chlorides and bromides when obtained by direct halogenation of aromatic compounds give a mixture of products that are difficult to separate. However, in the diazonium salt replacement method, a pure single product is formed. This also provides a good synthetic route for the preparation of aryl iodides and fluorides which are not obtained by direct halogenation.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




