
Which of the following methods is used to estimate the hardness of water?
(A). Conductivity method
(B). EDTA method
(C). Titrimetric method
(D). Distillation method
Answer
590.4k+ views
Hint: Hard water has a high amount of dissolved minerals like calcium and magnesium. Hard water is formed when water percolates through deposits of limestone, chalk or gypsum which are largely made up of calcium and magnesium salts of carbonates, bicarbonates and sulphates. Hardness is measured in milligrams of calcium carbonate present per litre.
Complete step by step answer:
The hardness of water is caused due to the presence of dissolved magnesium and calcium salts. However, due to the presence of dissolved calcium or magnesium sulphates, permanent hardness of water is caused because these salts cannot be removed by boiling. On the other hand, the dissolved magnesium and calcium bicarbonates cause temporary hardness in water as they can be removed by boiling the water.
The hard water buildup in pipes causes less water flow due to dropped pressure. The hard water also builds up on geysers and other elements and damages them as the heating efficiency of these elements get reduced and this will increase the consumption of electricity. Moreover, the hard water reduces the lathering properties of soaps and detergents, causing the more use of these products. There are many more disadvantages of using hard water.
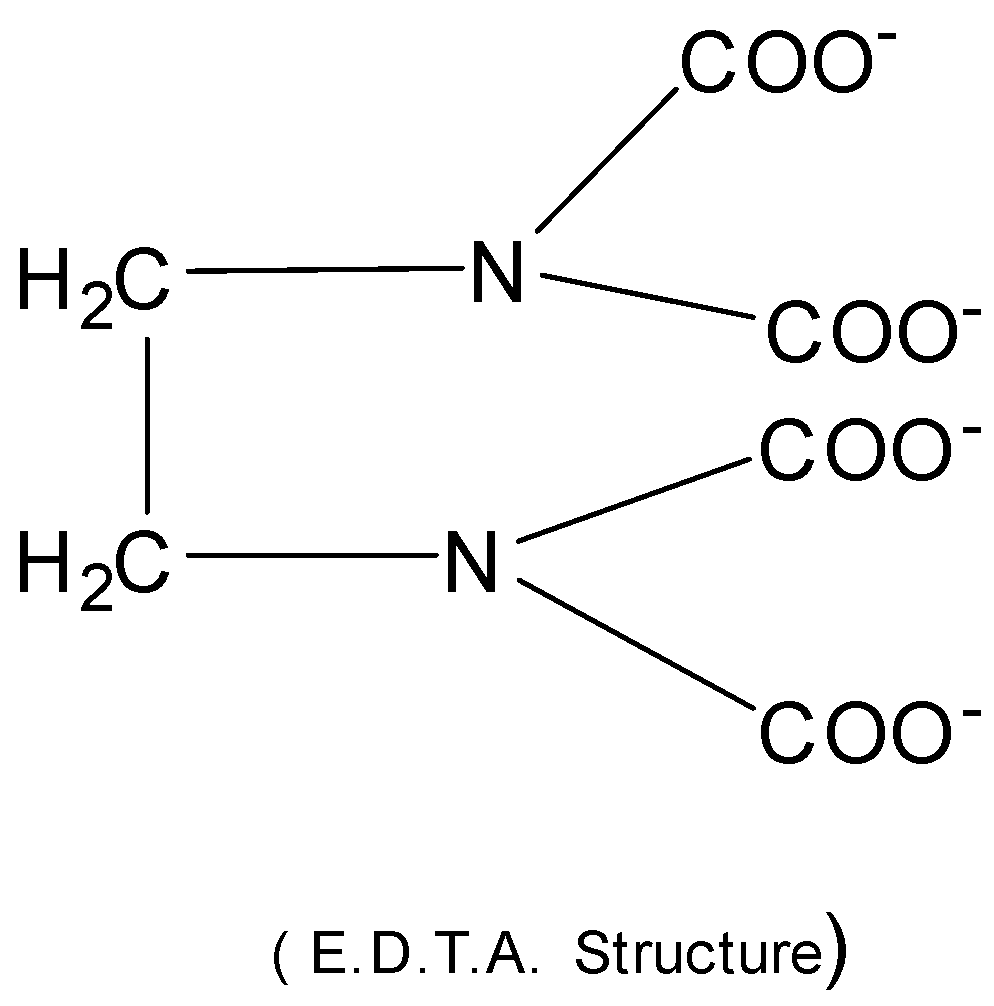
So, it is important to estimate the hardness of water so as to minimize the effect caused by hard water. The hardness of water can be determined by the complexometric titration using Ethylene diamine tetra acetic acid ( EDTA ) . EDTA in the form of its $di - $ sodium salt creates a complex with $C{a^{2 + }}$ and $M{g^{2 + }}$ ions of water. Then $C{a^{2 + }}$ and $M{g^{2 + }}$ ions form stable metal complexes with EDTA and the color changes from wine red to blue at the end point.
So, the correct answer is Option B.
Note:
EDTA is a multidentate ligand
It’s denticity (multiplicity) is $6.$
EDTA coordinates through $40$ atoms and $2N$ atoms.
Complete step by step answer:
The hardness of water is caused due to the presence of dissolved magnesium and calcium salts. However, due to the presence of dissolved calcium or magnesium sulphates, permanent hardness of water is caused because these salts cannot be removed by boiling. On the other hand, the dissolved magnesium and calcium bicarbonates cause temporary hardness in water as they can be removed by boiling the water.
The hard water buildup in pipes causes less water flow due to dropped pressure. The hard water also builds up on geysers and other elements and damages them as the heating efficiency of these elements get reduced and this will increase the consumption of electricity. Moreover, the hard water reduces the lathering properties of soaps and detergents, causing the more use of these products. There are many more disadvantages of using hard water.
So, it is important to estimate the hardness of water so as to minimize the effect caused by hard water. The hardness of water can be determined by the complexometric titration using Ethylene diamine tetra acetic acid ( EDTA ) . EDTA in the form of its $di - $ sodium salt creates a complex with $C{a^{2 + }}$ and $M{g^{2 + }}$ ions of water. Then $C{a^{2 + }}$ and $M{g^{2 + }}$ ions form stable metal complexes with EDTA and the color changes from wine red to blue at the end point.
So, the correct answer is Option B.
Note:
EDTA is a multidentate ligand
It’s denticity (multiplicity) is $6.$
EDTA coordinates through $40$ atoms and $2N$ atoms.

Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Give 10 examples of unisexual and bisexual flowers

Coming together federation is practiced in A India class 12 social science CBSE

How was the Civil Disobedience Movement different from class 12 social science CBSE




