Which of the following compounds will have the highest melting point?
A.Chlorobenzene
B.o-dichlorobenzene
C.m-dichlorobenzene
D.p-dichlorobenzene
Answer
613.5k+ views
Hint: The melting point is usually defined as the point at which the material changes from solid to a liquid. Moreover, melting points may be defined in various ways, each corresponding to a different residual amount of solid or liquid. The main criteria for melting point of such compounds is based on molecular packing.
Complete step by step answer:
Generally, the temperature at which the solid changes its state to liquid at atmospheric pressure is called the melting point of that liquid. Now, in case of these compounds like o-dichlorobenzene, m-dichlorobenzene and p-dichlorobenzene, the main criteria for determining the melting point is molecular packing. The melting point is directly proportional to the symmetry of the compound. We can say that the symmetry of the compound goes in the same manner as melting point.
So, p-dichlorobenzene is more symmetrical than the o- and m- isomers. Therefore, it fits more closely in the crystal lattice and hence, more energy is required to break the crystal lattice of p-dichlorobenzene. So, it is clear that p-dichlorobenzene will have the highest melting point than the o- and m- isomers. It has a melting point of ${53.5^ \circ }C$ . The three types of isomers are as shown:
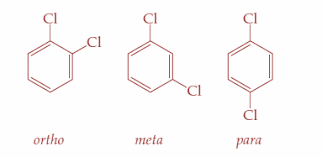
Hence, option D is correct.
Note: p-dichlorobenzene is used as a fumigant insecticide to control clothes moths. It is also found in deodorant blocks made for trash cans and toilets. Most of the people recognize the odor of this compound as the smell of mothballs. Further, acute inhalation or oral exposure to high concentrations may result in liver damage and may cause severe respiratory tract issues.
Complete step by step answer:
Generally, the temperature at which the solid changes its state to liquid at atmospheric pressure is called the melting point of that liquid. Now, in case of these compounds like o-dichlorobenzene, m-dichlorobenzene and p-dichlorobenzene, the main criteria for determining the melting point is molecular packing. The melting point is directly proportional to the symmetry of the compound. We can say that the symmetry of the compound goes in the same manner as melting point.
So, p-dichlorobenzene is more symmetrical than the o- and m- isomers. Therefore, it fits more closely in the crystal lattice and hence, more energy is required to break the crystal lattice of p-dichlorobenzene. So, it is clear that p-dichlorobenzene will have the highest melting point than the o- and m- isomers. It has a melting point of ${53.5^ \circ }C$ . The three types of isomers are as shown:

Hence, option D is correct.
Note: p-dichlorobenzene is used as a fumigant insecticide to control clothes moths. It is also found in deodorant blocks made for trash cans and toilets. Most of the people recognize the odor of this compound as the smell of mothballs. Further, acute inhalation or oral exposure to high concentrations may result in liver damage and may cause severe respiratory tract issues.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




