
Which of the following compounds show intramolecular hydrogen bonding?
A. o-nitrophenol
B. p-nitrophenol
C. phenol
D. salicylaldehyde
1. A and B
2. A and C
3. A and D
4. B and C
Answer
521.4k+ views
Hint: A special type of dipole-dipole interactions between hydrogen atoms and the highly electronegative elements i.e., fluorine, oxygen and nitrogen are known as hydrogen bonding. It is of two types, intermolecular hydrogen bonding and intramolecular hydrogen bonding.
Complete answer: Intramolecular hydrogen bonding: It is a type of hydrogen bonding in which the dipole-dipole interactions are observed within a single molecule. This usually occurs when two functional groups consisting of hydrogen atoms and an electronegative element are located very near to each other in a compound. Intramolecular hydrogen bonding is stronger than intermolecular hydrogen bonding because all the groups participating in hydrogen bonding are connected via covalent bond to the same molecule.
Among the given options,
o-nitrophenol: The structure of ortho nitrophenol is as follows:

As the nitro group and the hydroxyl group are present very near to each other i.e., at adjacent positions, so the hydrogen atom of hydroxyl group will form a dipole interaction with the oxygen atom of nitro group and hence, it will show intramolecular hydrogen bonding. The interactions will be as follows:
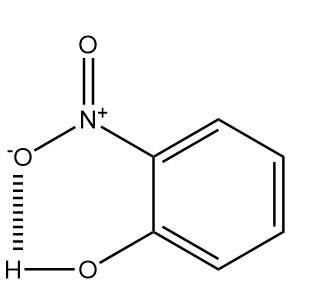
p-nitrophenol: The structure of para nitrophenol is as follows:
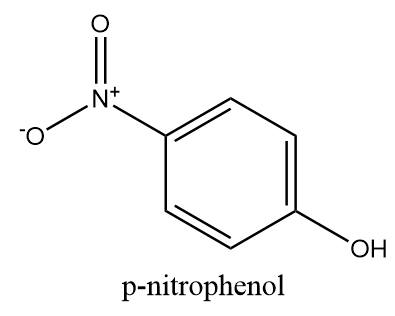
As the nitro group is present at the opposite position to that of hydroxyl group so there is no possibility of dipole-dipole interaction in the molecule and hence, it will not show intramolecular hydrogen bonding.
Phenol: The structure of phenol is as follows:

As there is no other functional group present with which it can show dipole interactions. So, phenol will not show intramolecular hydrogen bonding.
Salicylaldehyde: The structure of salicylaldehyde is as follows:

As the aldehyde group and the hydroxyl group are present very near to each other i.e., at adjacent positions, so the hydrogen atom of hydroxyl group will form a dipole interaction with the oxygen atom of aldehyde group and hence, it will show intramolecular hydrogen bonding. The interactions will be as follows:

Hence, only compounds A and D will show intramolecular hydrogen bonding. Thus, option (3) is the correct answer.
Note:
It is important to note that the hydrogen bonding will exist between those atoms which are not covalently bonded to each other. For example, in a water molecule, the hydrogen atom shows intermolecular hydrogen bonding with the oxygen atom of the other molecule of water, not within the same molecule.
Complete answer: Intramolecular hydrogen bonding: It is a type of hydrogen bonding in which the dipole-dipole interactions are observed within a single molecule. This usually occurs when two functional groups consisting of hydrogen atoms and an electronegative element are located very near to each other in a compound. Intramolecular hydrogen bonding is stronger than intermolecular hydrogen bonding because all the groups participating in hydrogen bonding are connected via covalent bond to the same molecule.
Among the given options,
o-nitrophenol: The structure of ortho nitrophenol is as follows:

As the nitro group and the hydroxyl group are present very near to each other i.e., at adjacent positions, so the hydrogen atom of hydroxyl group will form a dipole interaction with the oxygen atom of nitro group and hence, it will show intramolecular hydrogen bonding. The interactions will be as follows:

p-nitrophenol: The structure of para nitrophenol is as follows:

As the nitro group is present at the opposite position to that of hydroxyl group so there is no possibility of dipole-dipole interaction in the molecule and hence, it will not show intramolecular hydrogen bonding.
Phenol: The structure of phenol is as follows:

As there is no other functional group present with which it can show dipole interactions. So, phenol will not show intramolecular hydrogen bonding.
Salicylaldehyde: The structure of salicylaldehyde is as follows:

As the aldehyde group and the hydroxyl group are present very near to each other i.e., at adjacent positions, so the hydrogen atom of hydroxyl group will form a dipole interaction with the oxygen atom of aldehyde group and hence, it will show intramolecular hydrogen bonding. The interactions will be as follows:

Hence, only compounds A and D will show intramolecular hydrogen bonding. Thus, option (3) is the correct answer.
Note:
It is important to note that the hydrogen bonding will exist between those atoms which are not covalently bonded to each other. For example, in a water molecule, the hydrogen atom shows intermolecular hydrogen bonding with the oxygen atom of the other molecule of water, not within the same molecule.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Trending doubts
Draw a labelled sketch of the human eye class 12 physics CBSE

Which are the Top 10 Largest Countries of the World?

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Giving reasons state the signs positive or negative class 12 physics CBSE

Explain esterification reaction with the help of a class 12 chemistry CBSE

What is defined as a solenoid Depict a diagram with class 12 physics CBSE




