Which of the following compounds react with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] and \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\] to give alcohol/phenol?
A. \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]
B. \[{{\text{C}}_2}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]
C. \[{\text{C}}{{\text{H}}_3}{\text{NHC}}{{\text{H}}_3}\]
D. \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{NHC}}{{\text{H}}_{\text{3}}}\]
Answer
606.9k+ views
Hint: The reagent \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] and \[{\text{HCl}}\] used to distinguish primary, secondary and tertiary amines. Only primary aliphatic amines react with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] and \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\] . The product of the reaction is alcohol.
Complete Step by step answer: The reagent given to us is \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] and \[{\text{HCl}}\] and the reaction condition is \[{\text{0 - 4}}^\circ {\text{C}}\]. Only primary aliphatic amines react with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] and \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\] and give alcohol as the product.
The amine given in option A is \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]. Its structure is as follows:
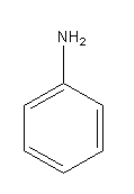
It is aromatic amine so it will not give phenol after reacting with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\].
Thus, option (A) \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\] is incorrect.
The amine given in option B is\[{{\text{C}}_2}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]. Its structure is as follows:
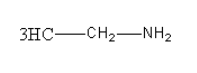
It is a primary aliphatic amine so it will give alcohol after reacting with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\].
So, option (B) \[{{\text{C}}_2}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]is correct.
The amine given in option C is \[{\text{C}}{{\text{H}}_3}{\text{NHC}}{{\text{H}}_3}\]. Its structure is as follows:
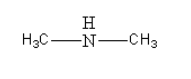
It is secondary aliphatic amine so it will not give alcohol after reacting with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\].
Thus, option (C) \[{\text{C}}{{\text{H}}_3}{\text{NHC}}{{\text{H}}_3}\] is incorrect.
The amine given in option D is \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{NHC}}{{\text{H}}_{\text{3}}}\]. Its structure is as follows:
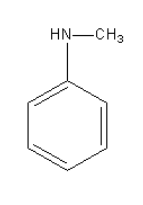
It is aromatic secondary amine so it will not give phenol after reacting with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\].
Thus, option (D) \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{NHC}}{{\text{H}}_{\text{3}}}\] is incorrect.
Hence, option (B) \[{{\text{C}}_2}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]is the correct answer.
Note: Aliphatic primary amines react with\[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\]and undergo diazotization to form alkane diazonium salt, which however being unstable decomposes to form a mixture of alcohols, alkene with the liberation of \[{{\text{N}}_{\text{2}}}\] gas. Secondary amines react with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] to form N-nitrosamines.
Complete Step by step answer: The reagent given to us is \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] and \[{\text{HCl}}\] and the reaction condition is \[{\text{0 - 4}}^\circ {\text{C}}\]. Only primary aliphatic amines react with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] and \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\] and give alcohol as the product.
The amine given in option A is \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]. Its structure is as follows:

It is aromatic amine so it will not give phenol after reacting with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\].
Thus, option (A) \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\] is incorrect.
The amine given in option B is\[{{\text{C}}_2}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]. Its structure is as follows:

It is a primary aliphatic amine so it will give alcohol after reacting with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\].
So, option (B) \[{{\text{C}}_2}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]is correct.
The amine given in option C is \[{\text{C}}{{\text{H}}_3}{\text{NHC}}{{\text{H}}_3}\]. Its structure is as follows:

It is secondary aliphatic amine so it will not give alcohol after reacting with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\].
Thus, option (C) \[{\text{C}}{{\text{H}}_3}{\text{NHC}}{{\text{H}}_3}\] is incorrect.
The amine given in option D is \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{NHC}}{{\text{H}}_{\text{3}}}\]. Its structure is as follows:

It is aromatic secondary amine so it will not give phenol after reacting with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\].
Thus, option (D) \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{NHC}}{{\text{H}}_{\text{3}}}\] is incorrect.
Hence, option (B) \[{{\text{C}}_2}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}\]is the correct answer.
Note: Aliphatic primary amines react with\[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] at \[{\text{0 - 4}}^\circ {\text{C}}\]and undergo diazotization to form alkane diazonium salt, which however being unstable decomposes to form a mixture of alcohols, alkene with the liberation of \[{{\text{N}}_{\text{2}}}\] gas. Secondary amines react with \[{\text{NaN}}{{\text{O}}_{\text{2}}}\] in \[{\text{HCl}}\] to form N-nitrosamines.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




