What is the formal charge of ${ NO }_{ 2 }^{ - }$?
Answer
630.3k+ views
Hint: The formal charge is the difference in the number of valence electrons in the atom and the number of valence electrons in the Lewis structure.
Complete answer step-by-step:
The formal charge of an atom in a molecule can be calculated by the following equation;
${ FC=V-N-B\div 2 }$
where V= number of valence electrons of the atom in the free atom
N = number of nonbonding electrons on this atom in the molecule
B = Total number of electrons shared in covalent bonds with other atoms in the molecule.
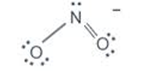
Nitrogen in the nitro group ${ NO }_{ 2 }^{ - }$ : FC= ${ 5-2-6\div 2=0 }$
Double bonded oxygen in ${ NO }_{ 2 }^{ - }$ : FC=${ 6-4-4\div 2=0 }$
Single bonded oxygen in ${ NO }_{ 2 }^{ - }$ : FC= ${ 6-6-2\div 2=-1 }$
Hence, ${ 0+0+(-1)=-1 }$ as expected for ${ NO }_{ 2 }^{ - }$
Additional Information:
Formal charge means ignore the lone pair of electrons and electrons shared by that atom from valence electrons then whatever is left with you is the significance of formal charge.
- It helps us to keep track of the electrons in a molecule.
- It tells you whether an atom has more electrons or protons associated with it.
- It allows us to assign electrons to a particular atom in a molecule.
Note: The possibility to make a mistake is that you may confuse between formal charge and the actual charge. But keep in mind that formal charges do not represent the actual charge on atoms in a molecule. The formal charges are assigned to the atoms in the Lewis structure to keep track of the electrons involved in the bonding.
Complete answer step-by-step:
The formal charge of an atom in a molecule can be calculated by the following equation;
${ FC=V-N-B\div 2 }$
where V= number of valence electrons of the atom in the free atom
N = number of nonbonding electrons on this atom in the molecule
B = Total number of electrons shared in covalent bonds with other atoms in the molecule.

Nitrogen in the nitro group ${ NO }_{ 2 }^{ - }$ : FC= ${ 5-2-6\div 2=0 }$
Double bonded oxygen in ${ NO }_{ 2 }^{ - }$ : FC=${ 6-4-4\div 2=0 }$
Single bonded oxygen in ${ NO }_{ 2 }^{ - }$ : FC= ${ 6-6-2\div 2=-1 }$
Hence, ${ 0+0+(-1)=-1 }$ as expected for ${ NO }_{ 2 }^{ - }$
Additional Information:
Formal charge means ignore the lone pair of electrons and electrons shared by that atom from valence electrons then whatever is left with you is the significance of formal charge.
- It helps us to keep track of the electrons in a molecule.
- It tells you whether an atom has more electrons or protons associated with it.
- It allows us to assign electrons to a particular atom in a molecule.
Note: The possibility to make a mistake is that you may confuse between formal charge and the actual charge. But keep in mind that formal charges do not represent the actual charge on atoms in a molecule. The formal charges are assigned to the atoms in the Lewis structure to keep track of the electrons involved in the bonding.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




