What is Boyle temperature?
Answer
627.9k+ views
Hint: Boyle temperature is described for real gases. Real gases do not adhere to the law of ideal gas and the particles of real gases have volumes.
Complete step-by-step solution:
Boyle temperature can be defined as the point in the temperature range in which a real gas starts to behave like an ideal gas at a pressure range. The temperature at which the second coefficient in the expression becomes zero is known as a Boyle temperature. This Boyle temperature balances out the attractive and the repulsive forces that are acting on a gas particle.
We can use the virial equation of state to calculate the Boyle temperature. Boyle temperature can be expressed in the terms of the virial coefficients.
$Z = 1 + \dfrac{B}{{{V_m}}} + ....$
Additional information:
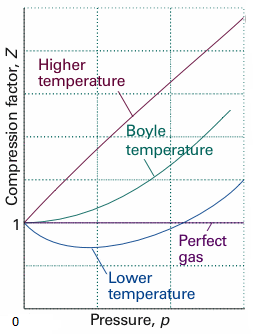
The above graph is plotted between the pressure $p$and compressibility factor$Z$. Boyle temperature is nicely marked in the graph. From this point, the real gas starts to behave like an ideal gas. The compression factor $Z$ is given as,
$Z = \dfrac{{p{V_m}}}{{RT}}$
Here, $Z$is the compressibility factor, ${V_m}$ is the volume,$R$ is the gas constant, $T$ is the
temperature and $p$ is the pressure.
$Z = 1$ for an ideal gas. Real gases show some deviation.
Note: Critical Temperature is different from Boyle's temperature. At the critical temperature, a gas shows non-ideal behavior. Critical temperature is lower than the Boyle temperature. At Boyle temperature a gas starts to behave like an ideal gas and for ideal gas, the compressibility factor is 1, that is, $Z = 1$
Complete step-by-step solution:
Boyle temperature can be defined as the point in the temperature range in which a real gas starts to behave like an ideal gas at a pressure range. The temperature at which the second coefficient in the expression becomes zero is known as a Boyle temperature. This Boyle temperature balances out the attractive and the repulsive forces that are acting on a gas particle.
We can use the virial equation of state to calculate the Boyle temperature. Boyle temperature can be expressed in the terms of the virial coefficients.
$Z = 1 + \dfrac{B}{{{V_m}}} + ....$
Additional information:

The above graph is plotted between the pressure $p$and compressibility factor$Z$. Boyle temperature is nicely marked in the graph. From this point, the real gas starts to behave like an ideal gas. The compression factor $Z$ is given as,
$Z = \dfrac{{p{V_m}}}{{RT}}$
Here, $Z$is the compressibility factor, ${V_m}$ is the volume,$R$ is the gas constant, $T$ is the
temperature and $p$ is the pressure.
$Z = 1$ for an ideal gas. Real gases show some deviation.
Note: Critical Temperature is different from Boyle's temperature. At the critical temperature, a gas shows non-ideal behavior. Critical temperature is lower than the Boyle temperature. At Boyle temperature a gas starts to behave like an ideal gas and for ideal gas, the compressibility factor is 1, that is, $Z = 1$
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




