What family is helium in?
Answer
561.3k+ views
Hint: The periodic table has all the elements arranged in accordance with their atomic numbers. The atomic number of elements leads to the electronic configuration of elements. Through electronic configuration, the elements can be classified according to the number of electrons in the orbitals.
Complete answer:
The modern periodic table consists of elements arranged in accordance with the modern periodic law that states, ‘the physical and chemical properties of elements are the periodic function of their atomic numbers’. This law clearly states that elements in the periodic table are arranged in order of their increasing atomic number.
In the periodic table, each element consists of an electron configuration that decides the nature and properties of that element. The atomic number is the number of electrons, and the electronic configuration is the filling of electrons into different orbitals, like, s, p, d, and f.
From the periodic table, we can tell the position of helium,
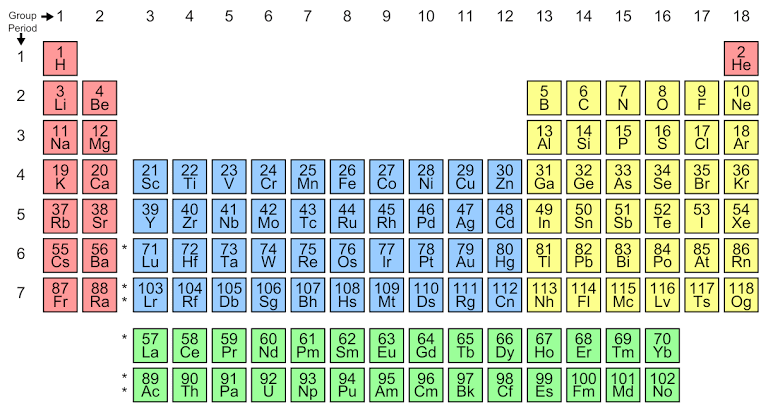
Helium, occupies, second number with 2 as the atomic number, so according to the Aufbau principle, the electron configuration of helium will be $He=1{{s}^{2}}$ , this shows it has a fully filled electron configuration, that makes it a member of the noble gases of group 18. The noble gases consist of all the orbitals as fully filled, and thus they are inert gases.
Hence, helium comes under the family of noble gases.
Note:
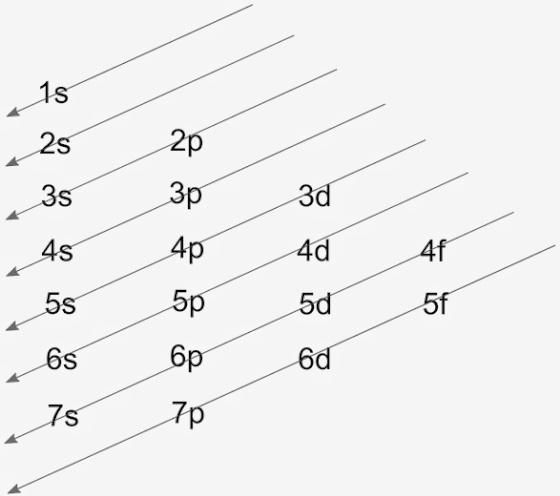
There are 5 families in the periodic table, which are, alkali metals, alkaline earth metals, transition metals, halogens, and noble gases. The filling of electrons takes place from the lower energy level to the higher energy levels according to the Aufbau principle, which is the name of the scientist. The filling is according to Aufbau diagram as:
Complete answer:
The modern periodic table consists of elements arranged in accordance with the modern periodic law that states, ‘the physical and chemical properties of elements are the periodic function of their atomic numbers’. This law clearly states that elements in the periodic table are arranged in order of their increasing atomic number.
In the periodic table, each element consists of an electron configuration that decides the nature and properties of that element. The atomic number is the number of electrons, and the electronic configuration is the filling of electrons into different orbitals, like, s, p, d, and f.
From the periodic table, we can tell the position of helium,

Helium, occupies, second number with 2 as the atomic number, so according to the Aufbau principle, the electron configuration of helium will be $He=1{{s}^{2}}$ , this shows it has a fully filled electron configuration, that makes it a member of the noble gases of group 18. The noble gases consist of all the orbitals as fully filled, and thus they are inert gases.
Hence, helium comes under the family of noble gases.
Note:
There are 5 families in the periodic table, which are, alkali metals, alkaline earth metals, transition metals, halogens, and noble gases. The filling of electrons takes place from the lower energy level to the higher energy levels according to the Aufbau principle, which is the name of the scientist. The filling is according to Aufbau diagram as:

Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is the full form of POSCO class 10 social science CBSE

Define Potential, Developed, Stock and Reserved resources

Which Country Has the Largest Border with India?

Complete the sentence with the most appropriate word class 10 english CBSE




