Two systems are in thermal equilibrium. The quantity which is common for them is
A. Heat
B. Momentum
C. specific heat
D. temperature
Answer
603.3k+ views
Hint: Zeroth law of thermodynamics states that two systems A and B, which are separately in thermal equilibrium with a third system C, are also in thermal equilibrium with each other. Any isolated system is in thermodynamic equilibrium.
Complete step by step answer:
Consider two systems A and B, separated by a wall that does not allow any exchange of energy between them. Such a wall is known as an insulating wall or adiabatic wall. The third system C is separated from system A and B by a conducting or diathermic wall.
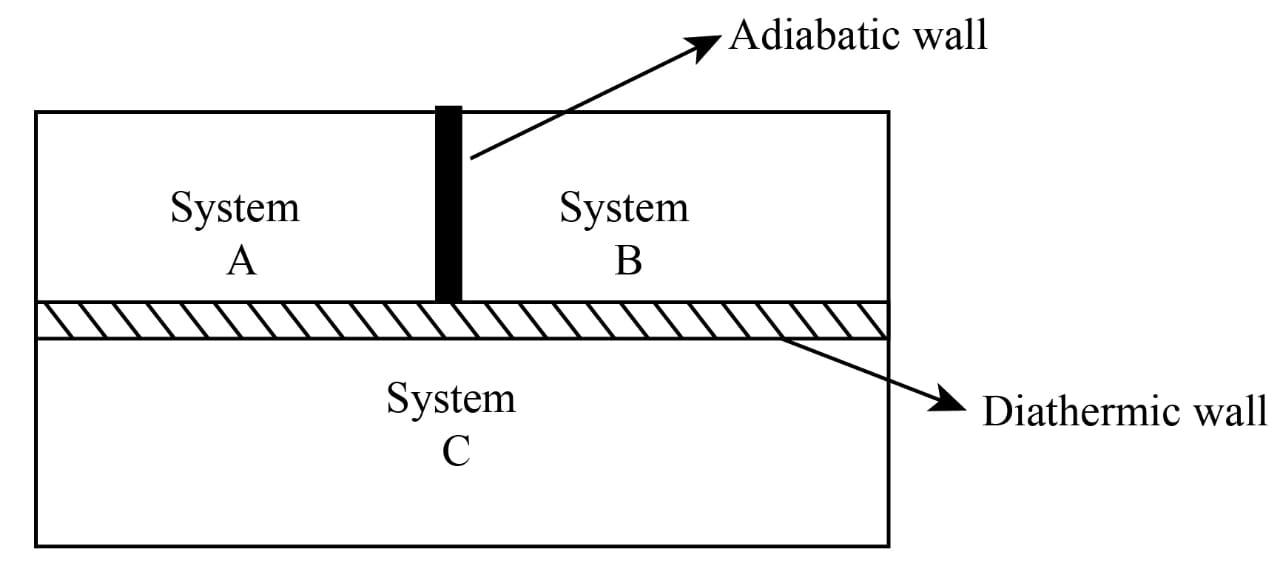
Since energy can be exchanged between the system A and C, so both system A and C are in thermal equilibrium. Similarly, energy can be exchanged between systems B and C. So both B and C are in thermal equilibrium. When the adiabatic wall between the systems A and B are removed, no energy transfer can take place between them because the temperature is the same; this shows that the two systems A and B are in thermal equilibrium; this observation leads to an important law known as Zeroth law of thermodynamics.
Hence, the correct option is (D).
Note:
Concept of temperature from Zeroth law of thermodynamics:
According to law, if system A is in thermal equilibrium with a system C,
Then the temperature of system A = Temperature of system C
Similarly, if system B is in thermal equilibrium with system C,
The temperature of system B = temperature of system C
Now, from the above relation, we conclude that,
The temperature of system A = Temperature of system B
Therefore, the temperature of a system is a physical quantity, equality of which is the only condition for thermal equilibrium.
Complete step by step answer:
Consider two systems A and B, separated by a wall that does not allow any exchange of energy between them. Such a wall is known as an insulating wall or adiabatic wall. The third system C is separated from system A and B by a conducting or diathermic wall.

Since energy can be exchanged between the system A and C, so both system A and C are in thermal equilibrium. Similarly, energy can be exchanged between systems B and C. So both B and C are in thermal equilibrium. When the adiabatic wall between the systems A and B are removed, no energy transfer can take place between them because the temperature is the same; this shows that the two systems A and B are in thermal equilibrium; this observation leads to an important law known as Zeroth law of thermodynamics.
Hence, the correct option is (D).
Note:
Concept of temperature from Zeroth law of thermodynamics:
According to law, if system A is in thermal equilibrium with a system C,
Then the temperature of system A = Temperature of system C
Similarly, if system B is in thermal equilibrium with system C,
The temperature of system B = temperature of system C
Now, from the above relation, we conclude that,
The temperature of system A = Temperature of system B
Therefore, the temperature of a system is a physical quantity, equality of which is the only condition for thermal equilibrium.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




