The surface tension of water is determined by capillary rise method in the laboratory. The principle involved is (symbols having their usual meaning):
A. $T=\dfrac{r}{3}\left(h+\dfrac{r}{3}\right)dg$
B.$T=\dfrac{r}{4}\left(h+\dfrac{r}{2}\right)dg$
C.$T=\dfrac{r}{2}\left(h+\dfrac{r}{3}\right)dg$
D.$T=\dfrac{r}{2}\left(\dfrac{h}{3}+r\right)dg$
Answer
548.7k+ views
Hint: We know that surface tension is the tendency of liquids to shrink or reduce the surface area of the liquid at the liquid air interface, due to the difference in the force of attraction of cohesion between the different interfaces present. And this is measured in SI units
Complete step-by-step solution:
To find or calculate the surface tension of liquids, we use the capillary rise method, where a thin tube called the capillary is introduced into the liquid, whose surface tension is to be studied.
This results in the rise of liquid inside the constrained spaces of the tube. The length of rise is proportional to the surface tension of the liquid.
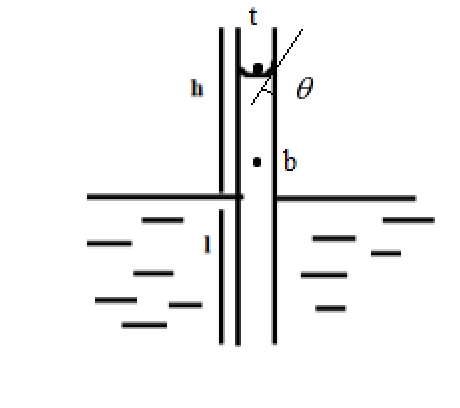
From Jurin’s law, we have, just above the liquid-air interface, the pressure experienced by the water molecules is equal to the atmospheric pressure.
$p_t=p_o+ p$
At any point inside the liquid, the pressure experienced by the liquid is less than the sum of the pressure due to the liquid and the pressure due to the atmosphere.
$p_b=p+p_l$
The pressure due to liquid depends on the height of liquid column $h$ above the point of interest and the density of the liquid $d$
$p_l=dgh$
Since there is an angle made at the liquid air interface, we have
$dgh=\dfrac{2\gamma cos \theta}{r}$ where, $\gamma$ is the surface tension due to contact angle $\theta$ and $r$ is the radius of the capillary tube.
Then, at any point in liquid, we have
$\gamma=\dfrac{r}{2cos\theta}dgh$
$\implies \gamma=\dfrac{r}{2}\left(\dfrac{h}{3}+r\right)dg$
Hence the answer is D. $T=\dfrac{r}{2}\left(\dfrac{h}{3}+r\right)dg$
Note: This is observed as the combined effect of the cohesive force acting between the liquid-liquid interface and the liquid –capillary. The difference between the attraction, results in the formation of meniscus, which tries to minimize the distance between the water molecules by attracting or pulling them closer to one another.
Complete step-by-step solution:
To find or calculate the surface tension of liquids, we use the capillary rise method, where a thin tube called the capillary is introduced into the liquid, whose surface tension is to be studied.
This results in the rise of liquid inside the constrained spaces of the tube. The length of rise is proportional to the surface tension of the liquid.

From Jurin’s law, we have, just above the liquid-air interface, the pressure experienced by the water molecules is equal to the atmospheric pressure.
$p_t=p_o+ p$
At any point inside the liquid, the pressure experienced by the liquid is less than the sum of the pressure due to the liquid and the pressure due to the atmosphere.
$p_b=p+p_l$
The pressure due to liquid depends on the height of liquid column $h$ above the point of interest and the density of the liquid $d$
$p_l=dgh$
Since there is an angle made at the liquid air interface, we have
$dgh=\dfrac{2\gamma cos \theta}{r}$ where, $\gamma$ is the surface tension due to contact angle $\theta$ and $r$ is the radius of the capillary tube.
Then, at any point in liquid, we have
$\gamma=\dfrac{r}{2cos\theta}dgh$
$\implies \gamma=\dfrac{r}{2}\left(\dfrac{h}{3}+r\right)dg$
Hence the answer is D. $T=\dfrac{r}{2}\left(\dfrac{h}{3}+r\right)dg$
Note: This is observed as the combined effect of the cohesive force acting between the liquid-liquid interface and the liquid –capillary. The difference between the attraction, results in the formation of meniscus, which tries to minimize the distance between the water molecules by attracting or pulling them closer to one another.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
How many bones are in the spine class 11 biology CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Draw a labelled diagram showing structure of LS of class 11 biology CBSE




