The structure of $Xe{{F}_{2}}$ involves hybridization of the type:
[A] $s{{p}^{3}}$
[B] $ds{{p}^{2}}$
[C] $s{{p}^{3}}d$
[D] $s{{p}^{3}}{{d}^{2}}$
Answer
613.8k+ views
Hint: To find the hybridisation of any complex, we need to determine the coordination number of the complex. If we know the coordination number, we can easily find out the hybridization and the geometry of the complex according to the valence bond theory.
Complete step by step answer:
In the valence bond theory, as we know atomic orbitals overlap with other atomic orbitals to form a molecule and thus creating new hybrid orbitals. This is known as the phenomenon of hybridisation.
The compound given to us is $Xe{{F}_{2}}$. To find its hybridisation, firstly let us write down its electronic configuration of the central metal atom that is xenon in this case.
Tellurium belongs to group 18 and its atomic number is 54. So, we can write its electronic configuration as- $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{10}}4{{s}^{2}}4{{p}^{6}}4{{d}^{10}}5{{s}^{2}}5{{p}^{6}}$.
We can see that there are 8 electrons in the valence shell. And from each fluorine atom we will have an electronic contribution of 1. So, from the two fluorine atoms we have a total contribution of 2.
So, the total number of electrons is 8 + 2 = 10. Or we can say that the number of electron pairs is 5.
Therefore, the coordination number is 5.
For coordination number 5, the corresponding hybridisation is $s{{p}^{3}}d$ and the shape is trigonal bipyramidal.
So, we can draw its structure as-
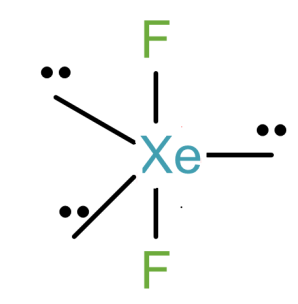
From this we can understand that the hybridisation is $s{{p}^{3}}d$
So, the correct answer is “Option C”.
Note: We can use the VSEPR theory which is the valence shell electron pair repulsion theory to correlate the hybridisation with the shape as well as the geometry of a molecule. Molecules settle in a shape where the electronic repulsion is the minimum and thus creates as lower energy shape as possible. Hybridization affects the bonds and the types of the bonds the molecule makes and thus is correlated to the shape of the molecule.
Complete step by step answer:
In the valence bond theory, as we know atomic orbitals overlap with other atomic orbitals to form a molecule and thus creating new hybrid orbitals. This is known as the phenomenon of hybridisation.
The compound given to us is $Xe{{F}_{2}}$. To find its hybridisation, firstly let us write down its electronic configuration of the central metal atom that is xenon in this case.
Tellurium belongs to group 18 and its atomic number is 54. So, we can write its electronic configuration as- $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{10}}4{{s}^{2}}4{{p}^{6}}4{{d}^{10}}5{{s}^{2}}5{{p}^{6}}$.
We can see that there are 8 electrons in the valence shell. And from each fluorine atom we will have an electronic contribution of 1. So, from the two fluorine atoms we have a total contribution of 2.
So, the total number of electrons is 8 + 2 = 10. Or we can say that the number of electron pairs is 5.
Therefore, the coordination number is 5.
For coordination number 5, the corresponding hybridisation is $s{{p}^{3}}d$ and the shape is trigonal bipyramidal.
So, we can draw its structure as-

From this we can understand that the hybridisation is $s{{p}^{3}}d$
So, the correct answer is “Option C”.
Note: We can use the VSEPR theory which is the valence shell electron pair repulsion theory to correlate the hybridisation with the shape as well as the geometry of a molecule. Molecules settle in a shape where the electronic repulsion is the minimum and thus creates as lower energy shape as possible. Hybridization affects the bonds and the types of the bonds the molecule makes and thus is correlated to the shape of the molecule.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




