The silicate anion in the mineral kinoite is a chain of three \[Si{O_4}^{4 - }\] tetrahedral that share corners with adjacent tetrahedral. The charge of the silicate anion is:
A.$ - 4$
B.$ - 8$
C.$ - 6$
D.$ - 2$
Answer
547.5k+ views
Hint: We have to know that the building block of all of these minerals is the silica tetrahedron, a combination of four oxygen atoms and one silicon atom. These are arranged such that planes drawn through the oxygen atoms form a tetrahedron.
Complete answer:
We have to know that the kinoite mineral has a molecular formula \[C{a_2}C{u_2}S{i_3}{O_8}{(OH)_4}\] it is a bluish colored copper silicate mineral. The crystal structure of this mineral is monoclinic crystal. It is lustrous in appearance and also somewhat transparent.
Now solving the question given it is given that kyanite mineral is a chain of three \[Si{O_4}^{4 - }\] tetrahedral that share corners with adjacent tetrahedral, thus first we need to draw what is asked that makes it easy to answer this question:
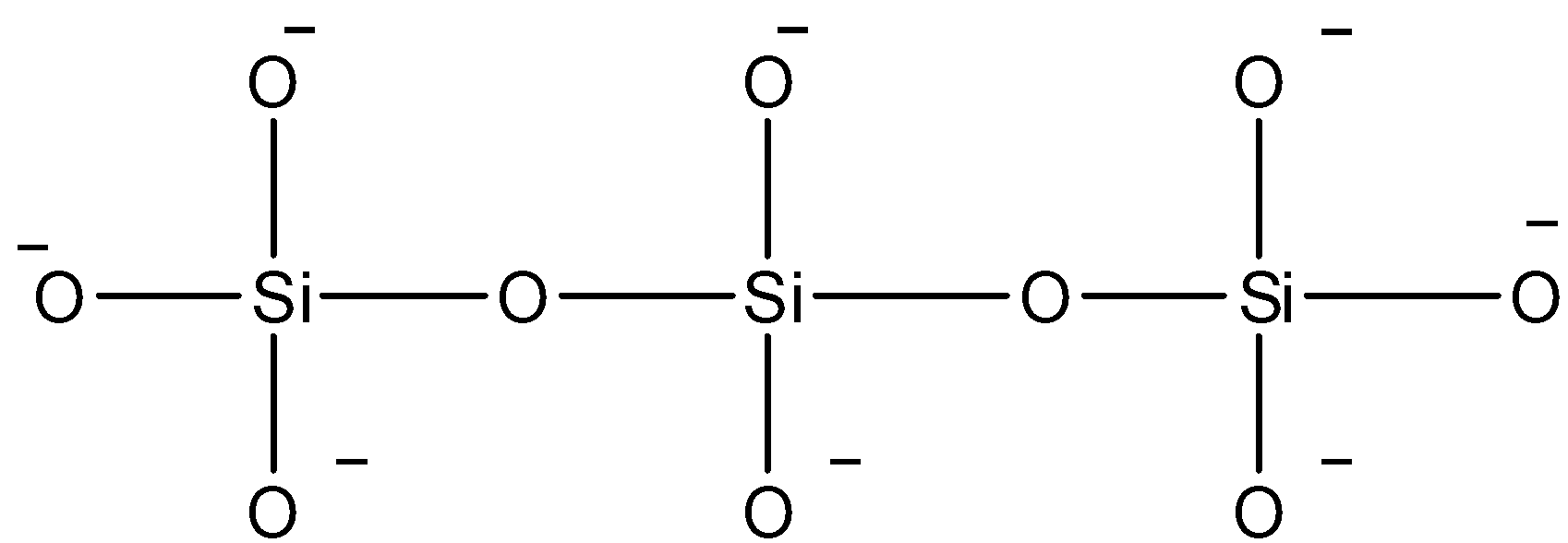
This is how the three \[Si{O_4}^{4 - }\] tetrahedral shares corners with adjacent tetrahedral. When we see at this molecule there are three silicon atoms bonded between two oxygen atoms and each silicon atom is carrying oxygen ion, thus the formula so formed is \[S{i_3}{O_{10}}^{8 - }\] . So we can conclude that the charge on this silicate ion is $ - 8$ .
From the above solved answer it is concluded that option B) is the correct option.
Note:
We have to know that the silicate is a family of atoms which is composed of silicon and oxygen atoms. It is anionic in nature as it carries negative charge on it. The general formula of silicates can be represented as\[{\left[ {Si{O^{\left( {4 - 2x} \right) - }}_{4 - x}} \right]_n}\],where \[0{\text{ }} \leqslant x < {\text{ }}2\].
The silicates include many classes like orthosilicates, pyrosilicates depending upon the changing values of \[x\] in the above equation.
Complete answer:
We have to know that the kinoite mineral has a molecular formula \[C{a_2}C{u_2}S{i_3}{O_8}{(OH)_4}\] it is a bluish colored copper silicate mineral. The crystal structure of this mineral is monoclinic crystal. It is lustrous in appearance and also somewhat transparent.
Now solving the question given it is given that kyanite mineral is a chain of three \[Si{O_4}^{4 - }\] tetrahedral that share corners with adjacent tetrahedral, thus first we need to draw what is asked that makes it easy to answer this question:

This is how the three \[Si{O_4}^{4 - }\] tetrahedral shares corners with adjacent tetrahedral. When we see at this molecule there are three silicon atoms bonded between two oxygen atoms and each silicon atom is carrying oxygen ion, thus the formula so formed is \[S{i_3}{O_{10}}^{8 - }\] . So we can conclude that the charge on this silicate ion is $ - 8$ .
From the above solved answer it is concluded that option B) is the correct option.
Note:
We have to know that the silicate is a family of atoms which is composed of silicon and oxygen atoms. It is anionic in nature as it carries negative charge on it. The general formula of silicates can be represented as\[{\left[ {Si{O^{\left( {4 - 2x} \right) - }}_{4 - x}} \right]_n}\],where \[0{\text{ }} \leqslant x < {\text{ }}2\].
The silicates include many classes like orthosilicates, pyrosilicates depending upon the changing values of \[x\] in the above equation.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




