The reaction of methyl magnesium iodide with acetone followed by hydrolysis, gives secondary alcohol.
a) True
b) False
Answer
580.2k+ views
Hint : In the metal – carbon bond the electronegativity of the alkyl group is maximum. Thus the alkyl group acts as a nucleophile and attacks the most electrophilic centre which in this case is a carbonyl group in acetone.
Complete Step By Step Answer:
Grignard reagents are the organometallic compound where the metal carbon bond is present. This method is the most convenient for the preparation of alcohols using the aldehydes and ketones.
In this reagent i.e. Grignard reagent (organomagnesium halide), the alkyl group acts as a nucleophile. As we know the carbonyl carbon is electron deficient due to the presence of electronegative atom oxygen, and acts as a good electrophilic centre.
When the organomagnesium halide attacks the electrophilic centre followed by hydrolysis, an additional product is obtained which results in the formation of alcohol.
Now there are some differences when we use Grignard reagent with aldehydes and with ketones. By selecting a proper Grignard reagent we can easily form primary, secondary and tertiary alcohols in reaction with aldehydes and ketones.
When formaldehyde reacts with methyl magnesium iodide, followed by hydrolysis, the formation of primary alcohol takes place.
When aldehydes other than formaldehyde reacts with methyl magnesium iodide, followed by hydrolysis, the formation of secondary alcohol takes place.
When methyl magnesium iodide reacts with acetone (ketone) followed by hydrolysis formation of tertiary alcohols takes place. And not the secondary alcohols. For e.g. in the reaction of methyl magnesium iodide with acetone which is a ketone and further followed by hydrolysis results in the formation of $ 2 - Methylpropan - 2 - ol $ that is a tertiary alcohol.
The mechanism can be drawn as follows-
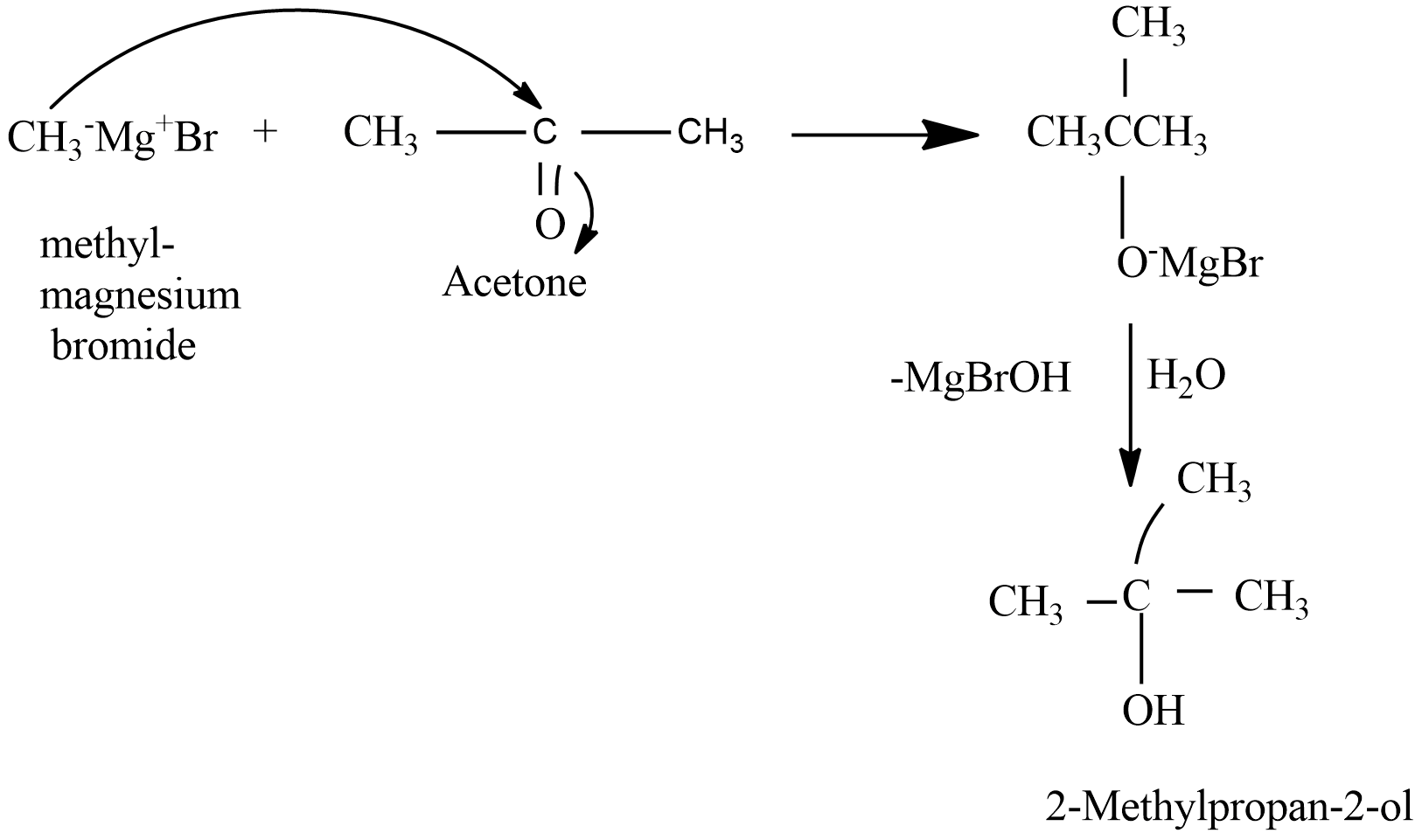
Hence the given statement is (b) False.
Note :
Alcohols can also be formed when the Grignard reagent reacts with epoxides. In this process ring opening of epoxide takes place followed by hydrolysis. Then the product can be obtained as alcohol. By this method with substituted epoxides we can form all primary, secondary and tertiary alcohols.
Complete Step By Step Answer:
Grignard reagents are the organometallic compound where the metal carbon bond is present. This method is the most convenient for the preparation of alcohols using the aldehydes and ketones.
In this reagent i.e. Grignard reagent (organomagnesium halide), the alkyl group acts as a nucleophile. As we know the carbonyl carbon is electron deficient due to the presence of electronegative atom oxygen, and acts as a good electrophilic centre.
When the organomagnesium halide attacks the electrophilic centre followed by hydrolysis, an additional product is obtained which results in the formation of alcohol.
Now there are some differences when we use Grignard reagent with aldehydes and with ketones. By selecting a proper Grignard reagent we can easily form primary, secondary and tertiary alcohols in reaction with aldehydes and ketones.
When formaldehyde reacts with methyl magnesium iodide, followed by hydrolysis, the formation of primary alcohol takes place.
When aldehydes other than formaldehyde reacts with methyl magnesium iodide, followed by hydrolysis, the formation of secondary alcohol takes place.
When methyl magnesium iodide reacts with acetone (ketone) followed by hydrolysis formation of tertiary alcohols takes place. And not the secondary alcohols. For e.g. in the reaction of methyl magnesium iodide with acetone which is a ketone and further followed by hydrolysis results in the formation of $ 2 - Methylpropan - 2 - ol $ that is a tertiary alcohol.
The mechanism can be drawn as follows-

Hence the given statement is (b) False.
Note :
Alcohols can also be formed when the Grignard reagent reacts with epoxides. In this process ring opening of epoxide takes place followed by hydrolysis. Then the product can be obtained as alcohol. By this method with substituted epoxides we can form all primary, secondary and tertiary alcohols.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




