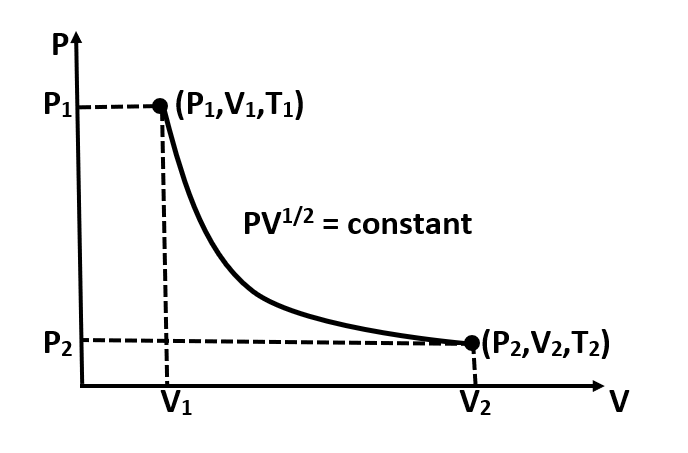
The P-V diagram of path followed by one mole of perfect gas in a cylindrical container is shown in figure, the work done when the gas is taken from state A to state B is:
$\begin{align}
& (A)2{{P}_{2}}{{V}_{1}}\left[ 1-\sqrt{\dfrac{{{V}_{2}}}{{{V}_{1}}}} \right] \\
& (B)2{{P}_{1}}{{V}_{1}}\left[ 1-\sqrt{\dfrac{{{V}_{1}}}{{{V}_{2}}}} \right] \\
& (C)2{{P}_{2}}{{V}_{2}}\left[ 1-\sqrt{\dfrac{{{V}_{1}}}{{{V}_{2}}}} \right] \\
& (D)2{{P}_{1}}{{V}_{2}}\left[ 1-\sqrt{\dfrac{{{V}_{1}}}{{{V}_{2}}}} \right] \\
\end{align}$

Answer
543.6k+ views
Hint: We have been given the condition that, $P{{V}^{\dfrac{1}{2}}}=k$ ,where k is any arbitrary constant. Now, this is an example of a polytropic process. So we will use the formula of work done in taking a gas from initial state to a final state of a polytropic process.
Complete answer:
It has been given in the question:
The initial parameters of gas are denoted by the following terms:
Initial volume : ${{V}_{1}}$
Initial pressure: ${{P}_{1}}$
Initial temperature: ${{T}_{1}}$
And, the final parameters of the gas are given by the following terms:
Final volume : ${{V}_{2}}$
Final pressure : ${{P}_{2}}$
Final temperature: ${{T}_{2}}$
Now, once we have defined the initial and final parameters of the gas, we can proceed ahead with calculating the work done in the given process.
As it is given:
$\Rightarrow P{{V}^{\dfrac{1}{2}}}=k$
This is a Polytropic process, with the value of :
$\Rightarrow n=\dfrac{1}{2}$
Now, the formula for work done in a polytropic process is given by the following equation:
$\Rightarrow W=\dfrac{{{P}_{1}}{{V}_{1}}-{{P}_{2}}{{V}_{2}}}{n-1}$
Putting the value of (n) in the above equation, we get:
$\begin{align}
& \Rightarrow W=\dfrac{{{P}_{1}}{{V}_{1}}-{{P}_{2}}{{V}_{2}}}{\dfrac{1}{2}-1} \\
& \Rightarrow W=2\left( {{P}_{2}}{{V}_{2}}-{{P}_{1}}{{V}_{1}} \right) \\
\end{align}$
Taking ${{P}_{2}}{{V}_{2}}$common out of the bracket in the right-hand side of the equation, we get the work done as:
$\Rightarrow W=2{{P}_{2}}{{V}_{2}}\left( 1-\dfrac{{{P}_{1}}{{V}_{1}}}{{{P}_{2}}{{V}_{2}}} \right)$ [Let this expression be equation number (1)]
Now, using the given polytropic function, we can write:
$\begin{align}
& \Rightarrow {{P}_{1}}{{({{V}_{1}})}^{\dfrac{1}{2}}}=k \\
& \Rightarrow {{P}_{1}}{{({{V}_{1}})}^{\dfrac{1}{2}}}\times {{(V)}^{\dfrac{1}{2}}}=k{{(V)}^{\dfrac{1}{2}}} \\
& \Rightarrow {{P}_{1}}{{V}_{1}}=k{{({{V}_{1}})}^{\dfrac{1}{2}}} \\
\end{align}$
Similarly, for the final state of gas we can write:
$\Rightarrow {{P}_{2}}{{V}_{2}}=k{{({{V}_{2}})}^{\dfrac{1}{2}}}$
Using these two equations in equation number (1), we get:
$\begin{align}
& \Rightarrow W=2{{P}_{2}}{{V}_{2}}\left( 1-\dfrac{k\sqrt{{{V}_{1}}}}{k\sqrt{{{V}_{2}}}} \right) \\
& \therefore W=2{{P}_{2}}{{V}_{2}}\left( 1-\dfrac{\sqrt{{{V}_{1}}}}{\sqrt{{{V}_{2}}}} \right) \\
\end{align}$
Hence, the work done in taking gas from $1\to 2$ under the given polytropic process is $2{{P}_{2}}{{V}_{2}}\left( 1-\sqrt{\dfrac{{{V}_{1}}}{{{V}_{2}}}} \right)$ .
So, the correct answer is “Option C”.
Note: In a polytropic process, (n) can take any value. If we keep on varying (n) we will see that for different values of (n), we get different processes. For example: for, $n=0$ , the process will become isobaric. For, $n=1$, the process will become isothermal, etc. Thus all the basic processes are a subset of the polytropic process.
Complete answer:
It has been given in the question:
The initial parameters of gas are denoted by the following terms:
Initial volume : ${{V}_{1}}$
Initial pressure: ${{P}_{1}}$
Initial temperature: ${{T}_{1}}$
And, the final parameters of the gas are given by the following terms:
Final volume : ${{V}_{2}}$
Final pressure : ${{P}_{2}}$
Final temperature: ${{T}_{2}}$
Now, once we have defined the initial and final parameters of the gas, we can proceed ahead with calculating the work done in the given process.
As it is given:
$\Rightarrow P{{V}^{\dfrac{1}{2}}}=k$
This is a Polytropic process, with the value of :
$\Rightarrow n=\dfrac{1}{2}$
Now, the formula for work done in a polytropic process is given by the following equation:
$\Rightarrow W=\dfrac{{{P}_{1}}{{V}_{1}}-{{P}_{2}}{{V}_{2}}}{n-1}$
Putting the value of (n) in the above equation, we get:
$\begin{align}
& \Rightarrow W=\dfrac{{{P}_{1}}{{V}_{1}}-{{P}_{2}}{{V}_{2}}}{\dfrac{1}{2}-1} \\
& \Rightarrow W=2\left( {{P}_{2}}{{V}_{2}}-{{P}_{1}}{{V}_{1}} \right) \\
\end{align}$
Taking ${{P}_{2}}{{V}_{2}}$common out of the bracket in the right-hand side of the equation, we get the work done as:
$\Rightarrow W=2{{P}_{2}}{{V}_{2}}\left( 1-\dfrac{{{P}_{1}}{{V}_{1}}}{{{P}_{2}}{{V}_{2}}} \right)$ [Let this expression be equation number (1)]
Now, using the given polytropic function, we can write:
$\begin{align}
& \Rightarrow {{P}_{1}}{{({{V}_{1}})}^{\dfrac{1}{2}}}=k \\
& \Rightarrow {{P}_{1}}{{({{V}_{1}})}^{\dfrac{1}{2}}}\times {{(V)}^{\dfrac{1}{2}}}=k{{(V)}^{\dfrac{1}{2}}} \\
& \Rightarrow {{P}_{1}}{{V}_{1}}=k{{({{V}_{1}})}^{\dfrac{1}{2}}} \\
\end{align}$
Similarly, for the final state of gas we can write:
$\Rightarrow {{P}_{2}}{{V}_{2}}=k{{({{V}_{2}})}^{\dfrac{1}{2}}}$
Using these two equations in equation number (1), we get:
$\begin{align}
& \Rightarrow W=2{{P}_{2}}{{V}_{2}}\left( 1-\dfrac{k\sqrt{{{V}_{1}}}}{k\sqrt{{{V}_{2}}}} \right) \\
& \therefore W=2{{P}_{2}}{{V}_{2}}\left( 1-\dfrac{\sqrt{{{V}_{1}}}}{\sqrt{{{V}_{2}}}} \right) \\
\end{align}$
Hence, the work done in taking gas from $1\to 2$ under the given polytropic process is $2{{P}_{2}}{{V}_{2}}\left( 1-\sqrt{\dfrac{{{V}_{1}}}{{{V}_{2}}}} \right)$ .
So, the correct answer is “Option C”.
Note: In a polytropic process, (n) can take any value. If we keep on varying (n) we will see that for different values of (n), we get different processes. For example: for, $n=0$ , the process will become isobaric. For, $n=1$, the process will become isothermal, etc. Thus all the basic processes are a subset of the polytropic process.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




