The pressure at the bottom of a tank of water is 3P, where P is the atmospheric pressure. If the water is drawn out until the level of water is lowered by one fifth, what is the pressure at the bottom of the tank now?
A. $ 2P $
B. $ \dfrac{{13}}{5}P $
C. $ \dfrac{8}{5}P $
D. $ \dfrac{4}{5}P $
Answer
602.7k+ views
Hint
The pressure at the bottom of a tank consists of two components. One is the atmospheric pressure and the other is the pressure exerted by the weight of the liquid.
$ P = \rho gh $ , Where, $ P $ is the total pressure at the bottom of a column of height $ h $ , filled with liquid of density $ \rho $ , and $ g $ is the gravitational acceleration. The SI unit of pressure is Pascal.
Complete step by step answer
We are provided with the information that the pressure at the bottom of a tank is $ 3P $ . We also know that the atmospheric pressure is given by $ P $ . We are aware that the total pressure at the bottom of a tank can be written as:
$ {P_t} = {P_{atm}} + {P_{liquid}} $
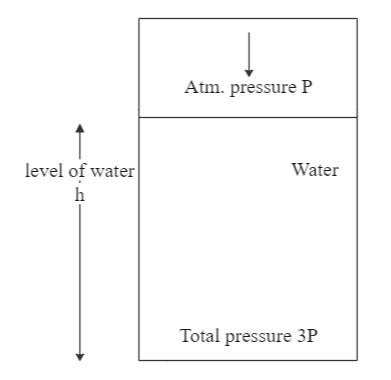
So, if we try to find the pressure exerted by the weight of water in this case, we get:
$ {P_w} = {P_t} - {P_{atm}}
\Rightarrow {P_w} = 3P - P $
This gives us the pressure due to the weight of water as $ 2P $ .
We also know that this pressure can be expressed as:
$ P = \rho gh $ [Eq. 1]
Now according to the question, we reduce the level of water (i.e. the height of water h) by one fifth. This gives us the new height as:
$h' = h - \dfrac{1}{5}h
\Rightarrow h' = \dfrac{{5h - h}}{5} $
Solving for $ h' $ , we get the new height = $ \dfrac{{4h}}{5} $
Hence, the new pressure will be given as:
$ {P_{new}} = \rho gh' $
Substituting the value of $ h' $ gives us:
${P_{new}} = \rho g \times \dfrac{4}{5}h
\Rightarrow {P_{new}} = \dfrac{4}{5}(\rho gh) $
We use Eq. 1 to get the answer in generic terms:
$ {P_{new}} = \dfrac{4}{5}P $
$ \therefore $ The correct answer is option D i.e. $ \dfrac{4}{5}P $.
Note
The trick here is to check the point at which we need to measure the pressure. For example, the pressure at the top of a tank would only be equal to the atmospheric pressure, but at the bottom it is different (as we saw in this question). The pressure in between is also calculated by the addition of atmospheric pressure and the pressure of the weight of the liquid above that height.
The pressure at the bottom of a tank consists of two components. One is the atmospheric pressure and the other is the pressure exerted by the weight of the liquid.
$ P = \rho gh $ , Where, $ P $ is the total pressure at the bottom of a column of height $ h $ , filled with liquid of density $ \rho $ , and $ g $ is the gravitational acceleration. The SI unit of pressure is Pascal.
Complete step by step answer
We are provided with the information that the pressure at the bottom of a tank is $ 3P $ . We also know that the atmospheric pressure is given by $ P $ . We are aware that the total pressure at the bottom of a tank can be written as:
$ {P_t} = {P_{atm}} + {P_{liquid}} $

So, if we try to find the pressure exerted by the weight of water in this case, we get:
$ {P_w} = {P_t} - {P_{atm}}
\Rightarrow {P_w} = 3P - P $
This gives us the pressure due to the weight of water as $ 2P $ .
We also know that this pressure can be expressed as:
$ P = \rho gh $ [Eq. 1]
Now according to the question, we reduce the level of water (i.e. the height of water h) by one fifth. This gives us the new height as:
$h' = h - \dfrac{1}{5}h
\Rightarrow h' = \dfrac{{5h - h}}{5} $
Solving for $ h' $ , we get the new height = $ \dfrac{{4h}}{5} $
Hence, the new pressure will be given as:
$ {P_{new}} = \rho gh' $
Substituting the value of $ h' $ gives us:
${P_{new}} = \rho g \times \dfrac{4}{5}h
\Rightarrow {P_{new}} = \dfrac{4}{5}(\rho gh) $
We use Eq. 1 to get the answer in generic terms:
$ {P_{new}} = \dfrac{4}{5}P $
$ \therefore $ The correct answer is option D i.e. $ \dfrac{4}{5}P $.
Note
The trick here is to check the point at which we need to measure the pressure. For example, the pressure at the top of a tank would only be equal to the atmospheric pressure, but at the bottom it is different (as we saw in this question). The pressure in between is also calculated by the addition of atmospheric pressure and the pressure of the weight of the liquid above that height.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




