The oxidation state of nitrogen in:
a.) \[{{N}_{3}}H\]
b.) \[N{{H}_{3}}\]
c.) \[N{{H}_{2}}OH\]
d.) \[{{N}_{2}}{{H}_{4}}\]
Answer
554.4k+ views
Hint: Nitrogen compounds exhibit oxidation states ranging from −3 (ammonia or amines) to +5 (nitric acid).
The electronic configuration of nitrogen is:\[1{{s}^{2}}2{{s}^{2}}2{{p}^{3}}\]
Nitrogen has 5 outer valence electrons, and the three electrons in 2p orbital are unpaired. Nitrogen can gain three electrons (into 2p orbital) or lose all the 5 valence electrons (from 2s and 2p orbitals) to obtain stable configuration.
Complete answer:
We have to calculate the oxidation states of nitrogen in the given compounds.
The given compounds are
\[{{N}_{3}}H\]- Hydrazoic acid.
\[N{{H}_{3}}\]- Ammonia
\[N{{H}_{2}}OH\]- Hydroxylamine
\[{{N}_{2}}{{H}_{4}}\]- Hydrazine
The oxidation state of nitrogen in \[{{N}_{3}}H\] is as follows.
There are three nitrogen atoms and one hydrogen atom. So,
3x+1 = 0
x = \[-\dfrac{1}{3}\]
The oxidation state of nitrogen in \[{{N}_{3}}H\]is \[-\dfrac{1}{3}\].
The oxidation state of nitrogen in \[N{{H}_{3}}\] is as follows.
There is one Nitrogen atom and three hydrogen atoms. So,
x+3(1) = 0
x+3 = 0
x = -3
The oxidation state of nitrogen in \[N{{H}_{3}}\]is -3.
The oxidation state of nitrogen in \[N{{H}_{2}}OH\] is as follows.
The structure of \[N{{H}_{2}}OH\] is
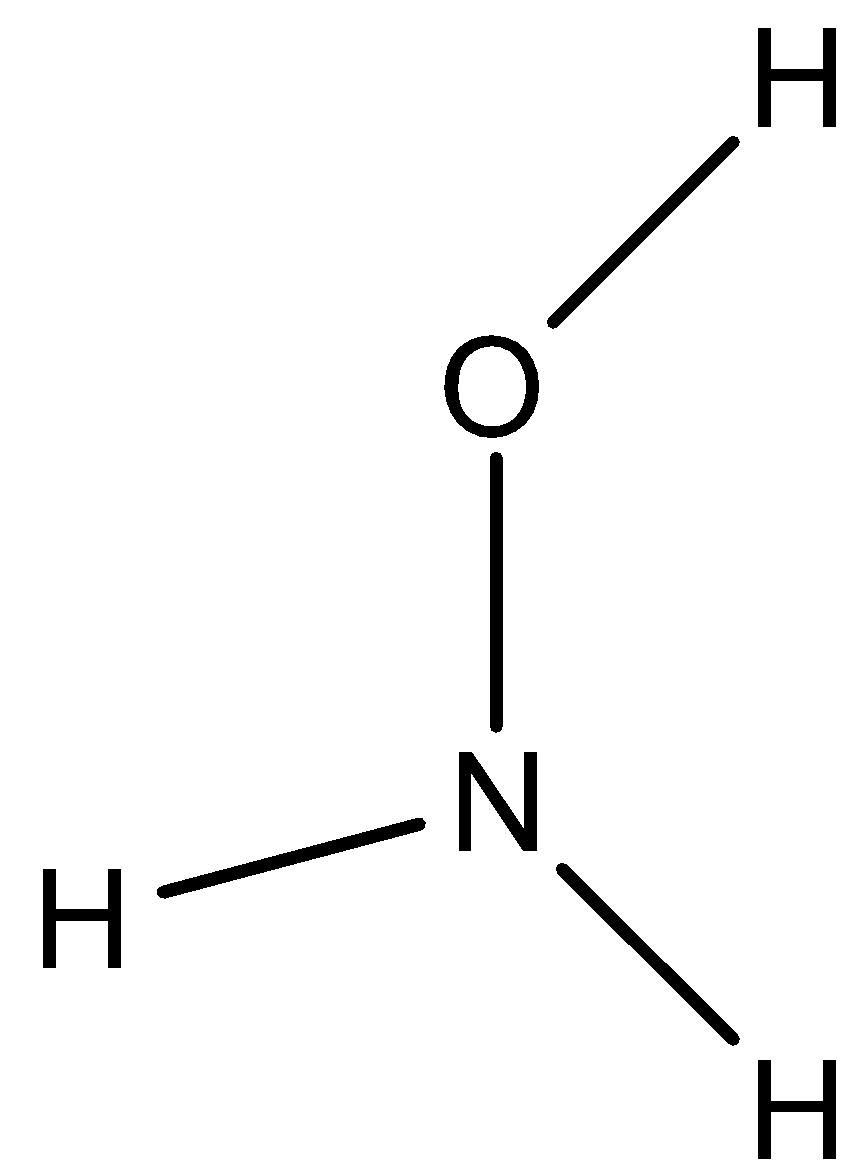
We know that there is one hydrogen atom, one oxygen atom and two types of hydrogen atoms.
One type of hydrogen atom (two hydrogens) directly attached to nitrogen and the second type of hydrogen attached to oxygen atom.
x + 3 (+1) – 2 = 0
x = -1
The oxidation state of nitrogen in \[N{{H}_{2}}OH\]is -1.
The oxidation state of nitrogen in \[{{N}_{2}}{{H}_{4}}\]is as follows.
There are two Nitrogen atoms and four hydrogen atoms.
2x + 4(1) = 0
2x = - 4
x = -2
The oxidation state of nitrogen in \[{{N}_{2}}{{H}_{4}}\] is -2.
Note: The various oxidation states of nitrogen are due to the presence of 5 valence electrons. It can donate 5 electrons or it can accept three electrons from other atoms to form stable molecules.
The electronic configuration of nitrogen is:\[1{{s}^{2}}2{{s}^{2}}2{{p}^{3}}\]
Nitrogen has 5 outer valence electrons, and the three electrons in 2p orbital are unpaired. Nitrogen can gain three electrons (into 2p orbital) or lose all the 5 valence electrons (from 2s and 2p orbitals) to obtain stable configuration.
Complete answer:
We have to calculate the oxidation states of nitrogen in the given compounds.
The given compounds are
\[{{N}_{3}}H\]- Hydrazoic acid.
\[N{{H}_{3}}\]- Ammonia
\[N{{H}_{2}}OH\]- Hydroxylamine
\[{{N}_{2}}{{H}_{4}}\]- Hydrazine
The oxidation state of nitrogen in \[{{N}_{3}}H\] is as follows.
There are three nitrogen atoms and one hydrogen atom. So,
3x+1 = 0
x = \[-\dfrac{1}{3}\]
The oxidation state of nitrogen in \[{{N}_{3}}H\]is \[-\dfrac{1}{3}\].
The oxidation state of nitrogen in \[N{{H}_{3}}\] is as follows.
There is one Nitrogen atom and three hydrogen atoms. So,
x+3(1) = 0
x+3 = 0
x = -3
The oxidation state of nitrogen in \[N{{H}_{3}}\]is -3.
The oxidation state of nitrogen in \[N{{H}_{2}}OH\] is as follows.
The structure of \[N{{H}_{2}}OH\] is

We know that there is one hydrogen atom, one oxygen atom and two types of hydrogen atoms.
One type of hydrogen atom (two hydrogens) directly attached to nitrogen and the second type of hydrogen attached to oxygen atom.
x + 3 (+1) – 2 = 0
x = -1
The oxidation state of nitrogen in \[N{{H}_{2}}OH\]is -1.
The oxidation state of nitrogen in \[{{N}_{2}}{{H}_{4}}\]is as follows.
There are two Nitrogen atoms and four hydrogen atoms.
2x + 4(1) = 0
2x = - 4
x = -2
The oxidation state of nitrogen in \[{{N}_{2}}{{H}_{4}}\] is -2.
Note: The various oxidation states of nitrogen are due to the presence of 5 valence electrons. It can donate 5 electrons or it can accept three electrons from other atoms to form stable molecules.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




