
The masses of two isotopes of chlorine are 34.980 and 36.978. If the radius of the circular path in Bainbridge spectrograph corresponding to lighter is 5 cm, the distance between the spots on photographic plate marked by the two isotopes will be
A. 5.7 cm
B. 0.57 cm
C. 0.57 mm
D. 0.57 m
Answer
600.9k+ views
Hint: Inside a spectrograph, particles move at a particular speed in a plane perpendicular to a magnetic field. Due to the action of the magnetic field the articles will move in a circle, we can use the Lorentz force formula to find the radius in which those particles move.
Formula used:
Lorentz force
F = q(v×B)
Complete step by step answer:
The Lorentz force will provide the necessary centripetal acceleration inside the spectrograph. Both the isotopes will have the same amount of charge and velocity on them. And as the direction of motion of particles and magnetic fields are perpendicular to each other, their cross product will be equal to their arithmetic product.
$\begin{align}
& \dfrac{m{{v}^{2}}}{r}=qvB \\
& \Rightarrow r=\dfrac{mv}{qB} \\
\end{align}$
So, the radius will be proportional to the mass of the particle in this case as all other quantities are the same for both the particles.
$\begin{align}
& \dfrac{r}{5}=\dfrac{36.978}{34.980} \\
& \Rightarrow r=5.285 \\
\end{align}$
As we can see the radius of the circular path in the Bainbridge spectrograph corresponding to heavier isotope will be 5.285 cm.
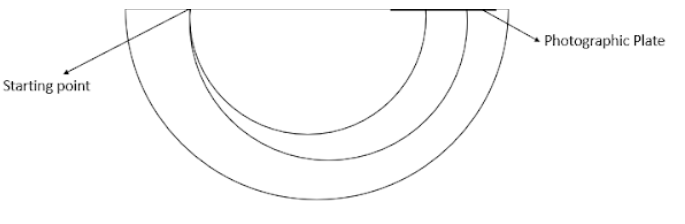
As we can see in the image, the distance between the two points on the photographic plate will be twice the difference of radii of the two circles. The difference between the radii is 0.285 cm and twice that will be 0.57 cm.
Hence, the correct option is B, i.e. 0.57 cm.
Note:
There can be a common mistake of not taking twice the difference of radii if the setup of the Bainbridge spectrograph is not visualized. The distance between the starting point and the point at which the particle strikes the plate is twice the radius made by the particle and both particles start at the same point.
Formula used:
Lorentz force
F = q(v×B)
Complete step by step answer:
The Lorentz force will provide the necessary centripetal acceleration inside the spectrograph. Both the isotopes will have the same amount of charge and velocity on them. And as the direction of motion of particles and magnetic fields are perpendicular to each other, their cross product will be equal to their arithmetic product.
$\begin{align}
& \dfrac{m{{v}^{2}}}{r}=qvB \\
& \Rightarrow r=\dfrac{mv}{qB} \\
\end{align}$
So, the radius will be proportional to the mass of the particle in this case as all other quantities are the same for both the particles.
$\begin{align}
& \dfrac{r}{5}=\dfrac{36.978}{34.980} \\
& \Rightarrow r=5.285 \\
\end{align}$
As we can see the radius of the circular path in the Bainbridge spectrograph corresponding to heavier isotope will be 5.285 cm.

As we can see in the image, the distance between the two points on the photographic plate will be twice the difference of radii of the two circles. The difference between the radii is 0.285 cm and twice that will be 0.57 cm.
Hence, the correct option is B, i.e. 0.57 cm.
Note:
There can be a common mistake of not taking twice the difference of radii if the setup of the Bainbridge spectrograph is not visualized. The distance between the starting point and the point at which the particle strikes the plate is twice the radius made by the particle and both particles start at the same point.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Giving reasons state the signs positive or negative class 12 physics CBSE

Explain esterification reaction with the help of a class 12 chemistry CBSE

What is defined as a solenoid Depict a diagram with class 12 physics CBSE




