The major product expected from the following reaction is:
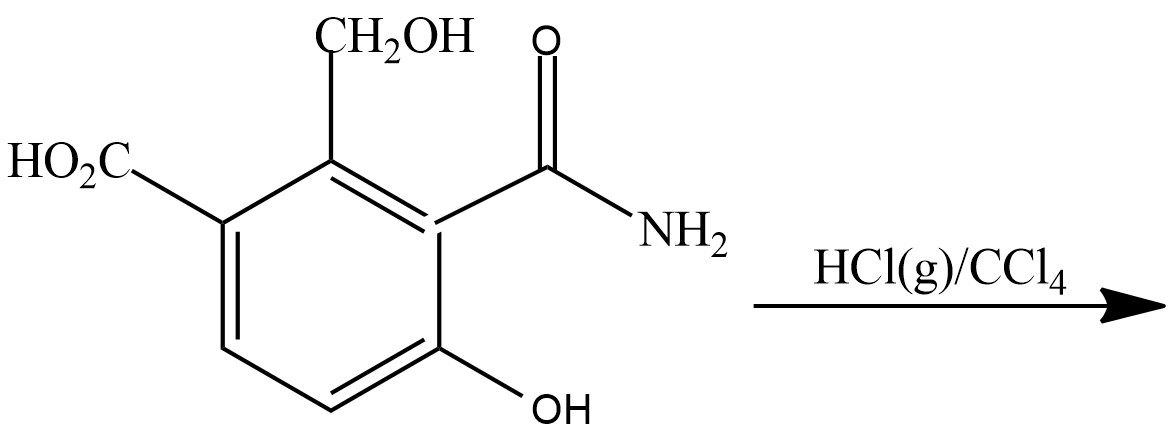
(A)
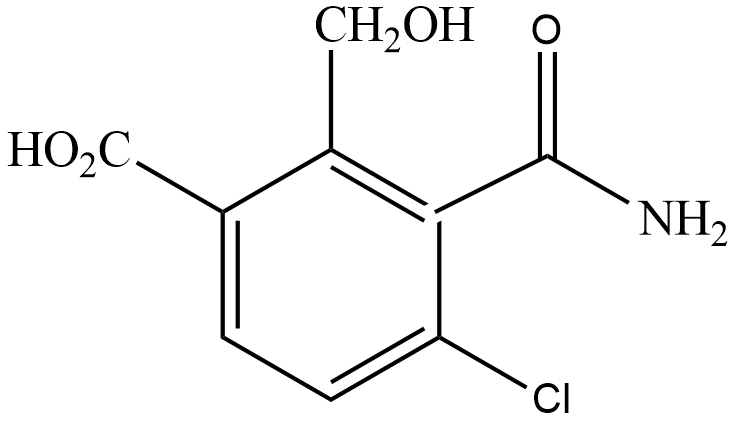
(B)
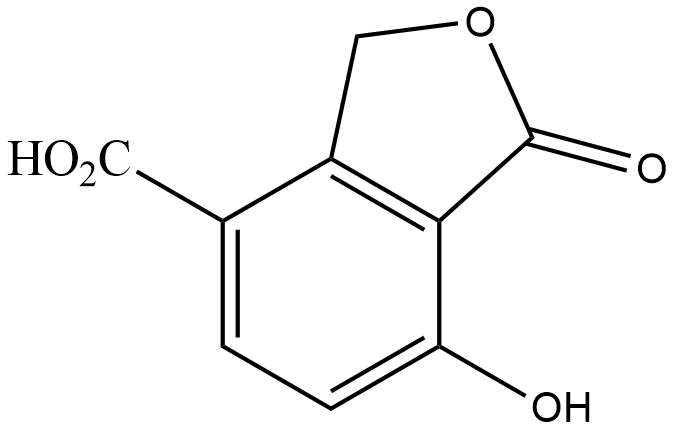
(C)
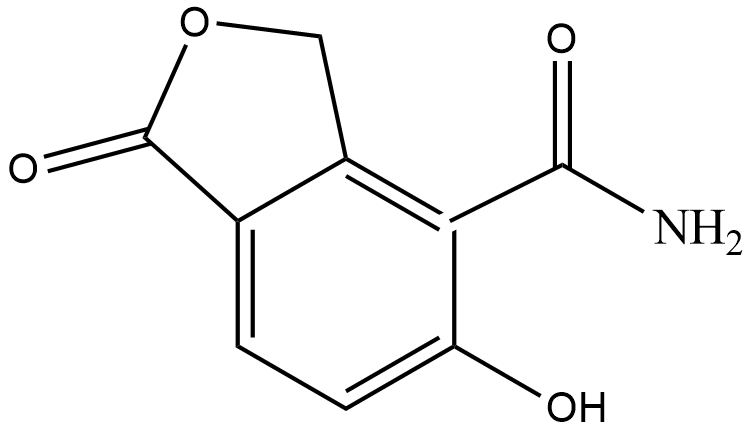
(D)
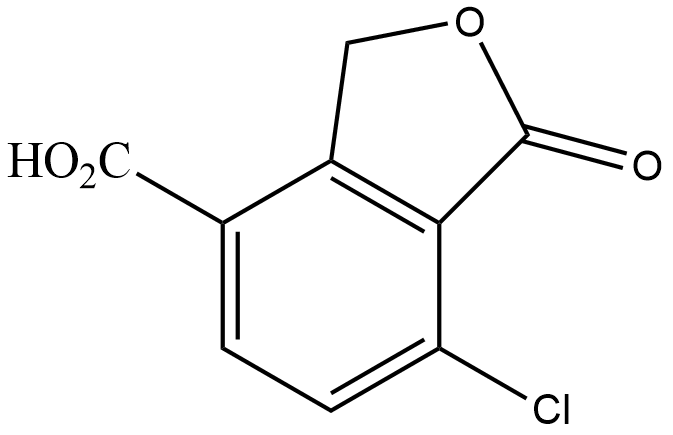





Answer
545.1k+ views
Hint : In the given compound, the functional groups carboxylic acid, methyl alcohol, amide, alcohol are attached to the benzene ring. The catalysts used here are concentrated hydrochloric acid $ HCl\; $ and Carbon tetrachloride $ CCl_4 $ is used as a solvent. These catalysts promote the dehydration reaction in the compound.
Complete Step By Step Answer:
The IUPAC name of the given compound is $ 2-methanol,3-amide,4-alcohol\;benzoic\;acid $
The catalyst given for the reaction is the Hydrochloric acid $ HCl\; $ in the gaseous state with carbon tetrachloride $ CCl_4 $ as a solvent.
The given conditions promote the esterification by the combination of carboxylic acid and alcohol.
Here, as the alcohol is not present separately in the solution, esterification of the methyl alcohol and the carboxylic acid will take place.
In the esterification reaction, first of all the hydrogen of the hydrochloric acid will get attracted to the double bond oxygen of carboxylic acid.
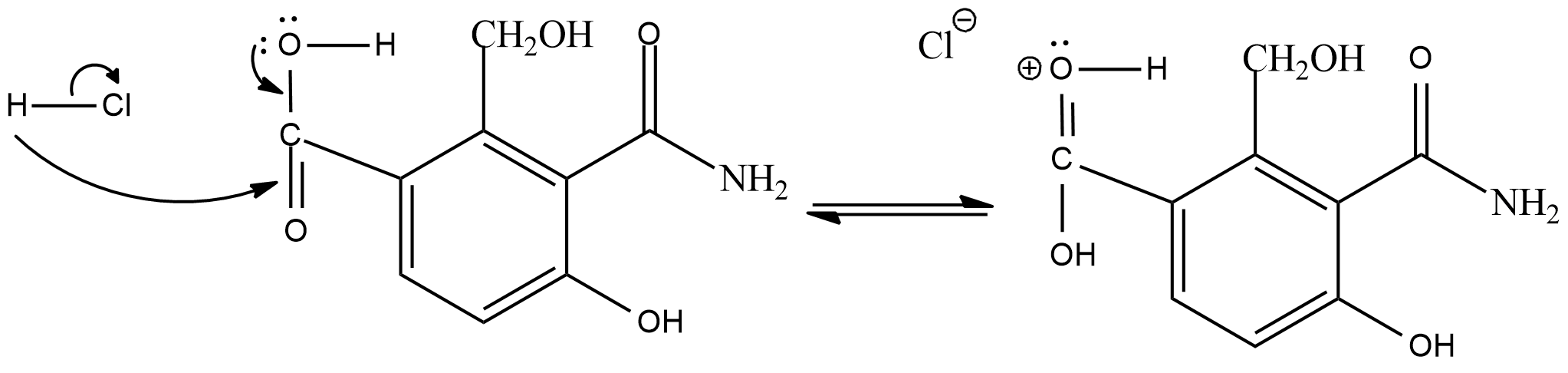
As the intermediate product will act as a electrophile, the adjacent alcohol group will act as a nucleophile and donate electrons
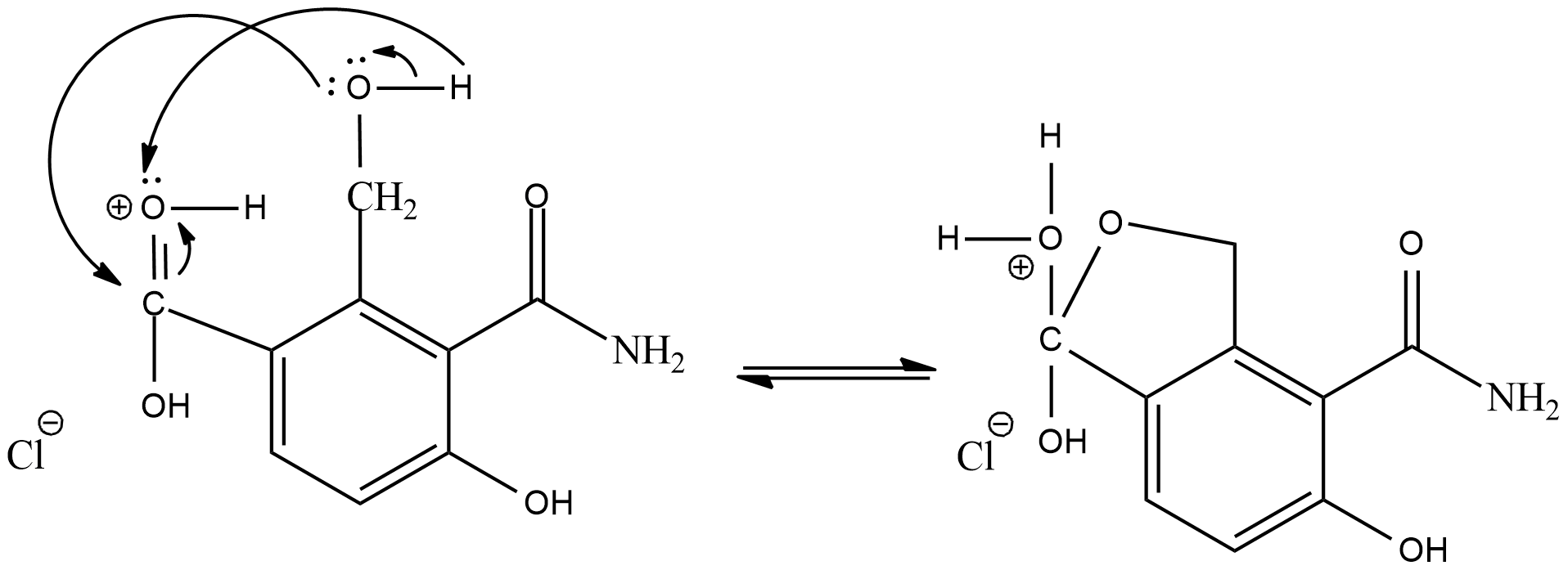
Now, the positive oxygen will leave the compound as water molecule and its valency will be balanced by the oxygen present as alcohol
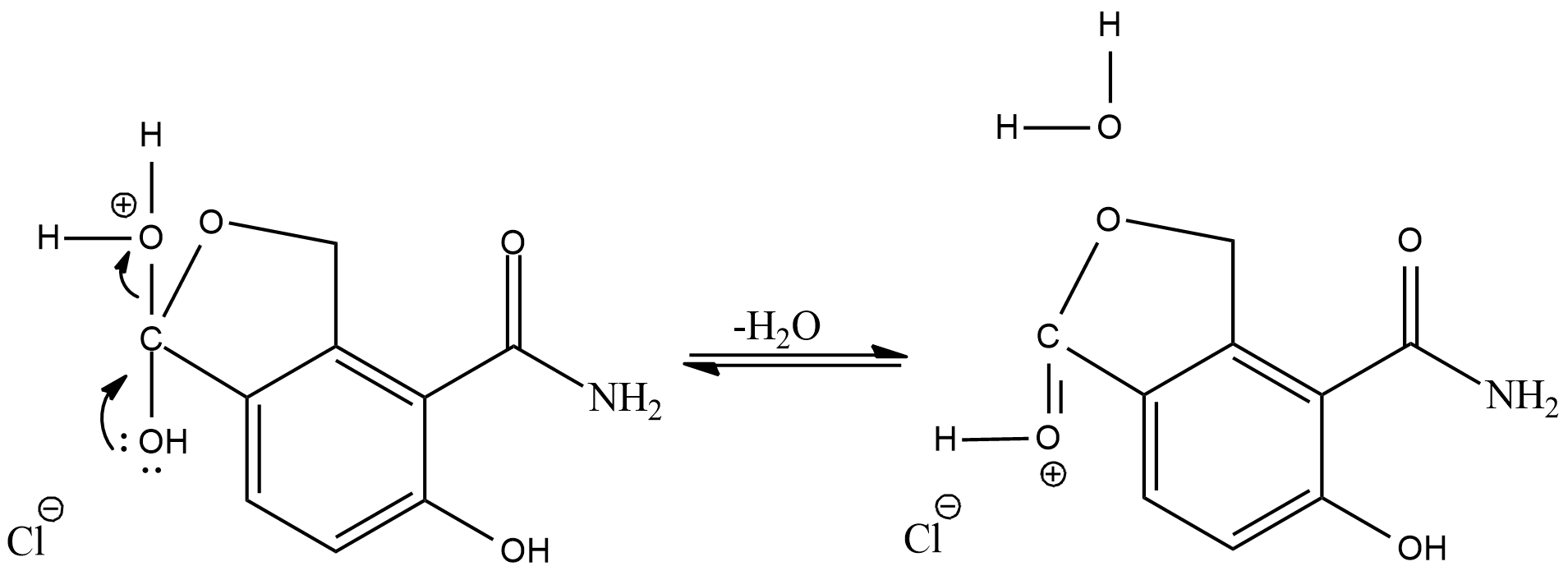
Now, the hydrogen added in the first step will again combine with chloride ion, and we will get a cyclic ester as the final product.
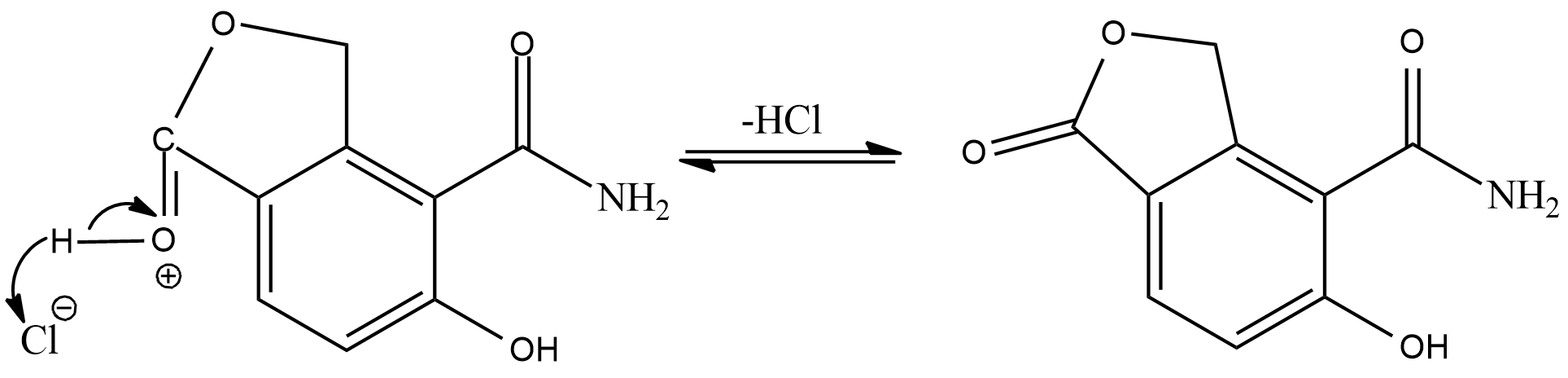
Hence, the correct answer is Option $ (C) $.
Note :
Alcohol groups show basic behavior, and on reaction with hydrochloric acid produce alkyl halides as salt. But this will not be possible in phenol, as the $ C-O $ bond possesses partial double bond nature due to resonance, and it is harder to break the bond. To convert the phenol to benzyl halide, we need to use $ SOCl_2 $ as a catalyst. Also, if we need to convert the amide group to ester, we need to use $ CeO_2 $ as a catalyst, which is not present here and hence, esterification of amide will not take place.
Complete Step By Step Answer:
The IUPAC name of the given compound is $ 2-methanol,3-amide,4-alcohol\;benzoic\;acid $
The catalyst given for the reaction is the Hydrochloric acid $ HCl\; $ in the gaseous state with carbon tetrachloride $ CCl_4 $ as a solvent.
The given conditions promote the esterification by the combination of carboxylic acid and alcohol.
Here, as the alcohol is not present separately in the solution, esterification of the methyl alcohol and the carboxylic acid will take place.
In the esterification reaction, first of all the hydrogen of the hydrochloric acid will get attracted to the double bond oxygen of carboxylic acid.

As the intermediate product will act as a electrophile, the adjacent alcohol group will act as a nucleophile and donate electrons

Now, the positive oxygen will leave the compound as water molecule and its valency will be balanced by the oxygen present as alcohol

Now, the hydrogen added in the first step will again combine with chloride ion, and we will get a cyclic ester as the final product.
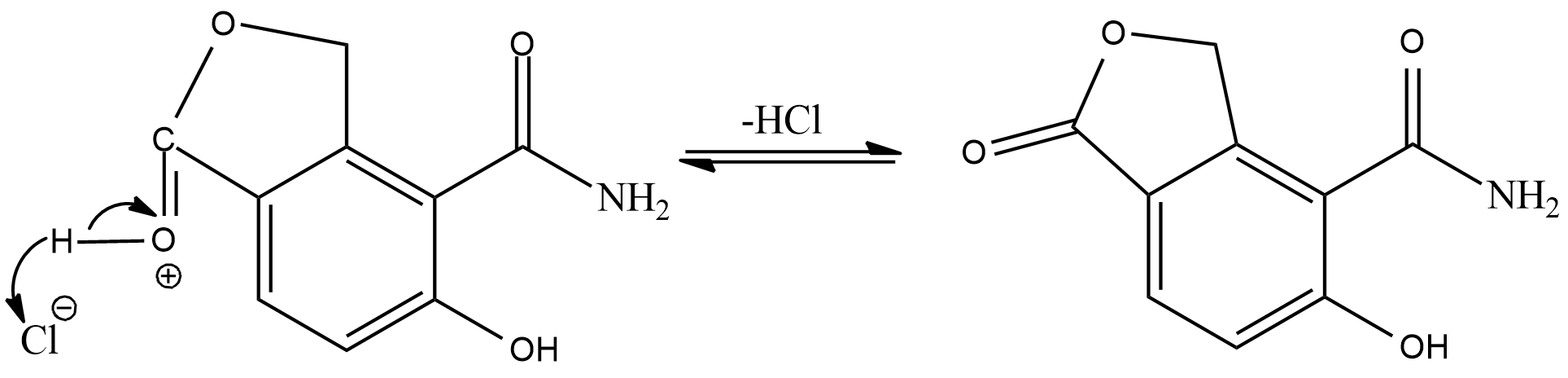
Hence, the correct answer is Option $ (C) $.
Note :
Alcohol groups show basic behavior, and on reaction with hydrochloric acid produce alkyl halides as salt. But this will not be possible in phenol, as the $ C-O $ bond possesses partial double bond nature due to resonance, and it is harder to break the bond. To convert the phenol to benzyl halide, we need to use $ SOCl_2 $ as a catalyst. Also, if we need to convert the amide group to ester, we need to use $ CeO_2 $ as a catalyst, which is not present here and hence, esterification of amide will not take place.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




