The length of carbon-carbon single bond of the compounds
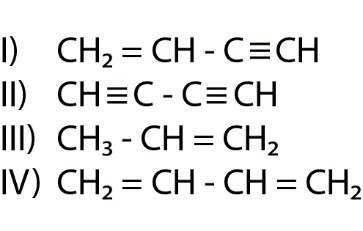
is expected to increase in the order
(A) Ⅲ > Ⅱ > Ⅰ > Ⅳ
(B)Ⅰ > Ⅲ > Ⅱ > Ⅳ
(C) Ⅲ > Ⅳ > Ⅰ > Ⅱ
(D)Ⅱ > Ⅳ > Ⅰ > Ⅲ
Answer
573.9k+ views
Hint: The length of the carbon-carbon single bond depends upon the hybridization of the carbon atom. The hybridization of the carbon contains S and P characters. More will be the p percentage, more will be the length of the carbon-carbon single bond.
Complete step by step solution:
For comparing the length of the carbon-carbon single bond, we need to check the hybridization of the carbons bonded via single bonds in each option.
In $ sp $ , $ 50\% $ S and $ 50\% $ P character is there.
In $ s{p^2} $ , $ 33.33\% $ S and $ 66.66\% $ P character is there.
In $ s{p^3} $ , $ 25\% $ S and $ 75\% $ P character is there.
Checking each option separately,
(Ⅰ)
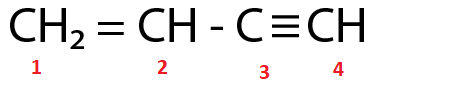
$ {C_2} $ and $ {C_3} $ are $ s{p^2} $ and $ sp $ hybridized respectively.
(Ⅱ)
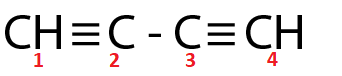
Both $ {C_2} $ and $ {C_3} $ are $ sp $ hybridized respectively.
(Ⅲ)
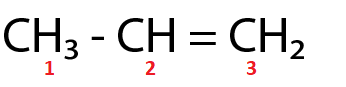
$ {C_1} $ and $ {C_2} $ are $ s{p^3} $ and $ s{p^2} $ hybridized respectively.
(Ⅳ)
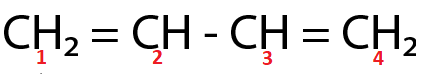 $ \because $
$ \because $
Here, both $ {C_2} $ and $ {C_3} $ are $ s{p^2} $ hybridized respectively.
Now, we know as the p character increases, the bond length of the carbon-carbon single bond increases.
Combining the percentage p character of the carbon-carbon single bond,
For the molecule in option (Ⅰ),
Total p percentage of carbons bonded via single bond = $ 66.66\% + 50\% = 116.66\% $
( $ \because $ $ {C_2} $ and $ {C_3} $ are $ s{p^2} $ and $ sp $ hybridized respectively.)
For the molecule in option (Ⅱ),
Total p percentage of carbons bonded via single bond = $ 50\% + 50\% = 100\% $
( $ \because $ $ {C_2} $ and $ {C_3} $ are $ sp $ hybridized respectively.)
For the molecule in option (Ⅲ),
Total p percentage of carbons bonded via single bond = $ 75\% + 66.66\% = 141.66\% $
( $ \because $ $ {C_1} $ and $ {C_2} $ are $ s{p^3} $ and $ s{p^2} $ hybridized respectively.)
For the molecule present in option (Ⅳ),
Total p percentage of carbons bonded via single bond = $ 66.66\% + 66.66\% = 133.32\% $
( $ \because $ $ {C_2} $ and $ {C_3} $ are $ s{p^2} $ hybridized respectively.)
Now, we can easily compare on the basis of combined percentage p character.
The molecule in option (Ⅲ) will have the longest carbon-carbon single bond length followed by (Ⅳ),
(Ⅰ) and (Ⅱ).
Therefore, the correct order will be Ⅲ > Ⅳ > Ⅰ > Ⅱ
So, the correct option will be option C: Ⅲ > Ⅳ > Ⅰ > Ⅱ.
Additional information:
A carbon-carbon bond is a covalent bond between two carbon atoms. Single bond is the most common form: a bond composed of two electrons, one from each of the two atoms. The carbon-carbon single bond is a sigma bond and is formed between one hybridized orbital from each of the carbon atoms.
Note:
The carbon-carbon single bond length is directly proportional to the combined percentage of p of both the carbon atoms bonded via single bond and inversely proportional to the combined percentage of s of both the carbon atoms bonded via single bond.
As the combined percentage of p increases, bond length of carbon-carbon single bond increases.
Complete step by step solution:
For comparing the length of the carbon-carbon single bond, we need to check the hybridization of the carbons bonded via single bonds in each option.
In $ sp $ , $ 50\% $ S and $ 50\% $ P character is there.
In $ s{p^2} $ , $ 33.33\% $ S and $ 66.66\% $ P character is there.
In $ s{p^3} $ , $ 25\% $ S and $ 75\% $ P character is there.
Checking each option separately,
(Ⅰ)
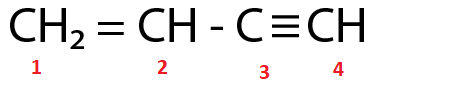
$ {C_2} $ and $ {C_3} $ are $ s{p^2} $ and $ sp $ hybridized respectively.
(Ⅱ)
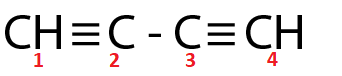
Both $ {C_2} $ and $ {C_3} $ are $ sp $ hybridized respectively.
(Ⅲ)
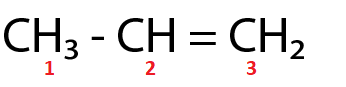
$ {C_1} $ and $ {C_2} $ are $ s{p^3} $ and $ s{p^2} $ hybridized respectively.
(Ⅳ)
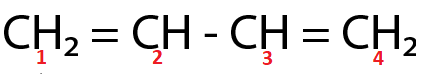
Here, both $ {C_2} $ and $ {C_3} $ are $ s{p^2} $ hybridized respectively.
Now, we know as the p character increases, the bond length of the carbon-carbon single bond increases.
Combining the percentage p character of the carbon-carbon single bond,
For the molecule in option (Ⅰ),
Total p percentage of carbons bonded via single bond = $ 66.66\% + 50\% = 116.66\% $
( $ \because $ $ {C_2} $ and $ {C_3} $ are $ s{p^2} $ and $ sp $ hybridized respectively.)
For the molecule in option (Ⅱ),
Total p percentage of carbons bonded via single bond = $ 50\% + 50\% = 100\% $
( $ \because $ $ {C_2} $ and $ {C_3} $ are $ sp $ hybridized respectively.)
For the molecule in option (Ⅲ),
Total p percentage of carbons bonded via single bond = $ 75\% + 66.66\% = 141.66\% $
( $ \because $ $ {C_1} $ and $ {C_2} $ are $ s{p^3} $ and $ s{p^2} $ hybridized respectively.)
For the molecule present in option (Ⅳ),
Total p percentage of carbons bonded via single bond = $ 66.66\% + 66.66\% = 133.32\% $
( $ \because $ $ {C_2} $ and $ {C_3} $ are $ s{p^2} $ hybridized respectively.)
Now, we can easily compare on the basis of combined percentage p character.
The molecule in option (Ⅲ) will have the longest carbon-carbon single bond length followed by (Ⅳ),
(Ⅰ) and (Ⅱ).
Therefore, the correct order will be Ⅲ > Ⅳ > Ⅰ > Ⅱ
So, the correct option will be option C: Ⅲ > Ⅳ > Ⅰ > Ⅱ.
Additional information:
A carbon-carbon bond is a covalent bond between two carbon atoms. Single bond is the most common form: a bond composed of two electrons, one from each of the two atoms. The carbon-carbon single bond is a sigma bond and is formed between one hybridized orbital from each of the carbon atoms.
Note:
The carbon-carbon single bond length is directly proportional to the combined percentage of p of both the carbon atoms bonded via single bond and inversely proportional to the combined percentage of s of both the carbon atoms bonded via single bond.
As the combined percentage of p increases, bond length of carbon-carbon single bond increases.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




