The IUPAC name of sec.butyl alcohol is:
A. 1-butanol
B. 2-butanol
C. 2-methyl-1-butanol
D. 2-methyl-2-butanol
Answer
592.2k+ views
Hint: The prefixes that is sec (from “secondary”), tert (from “tertiary”) etc. denotes the position of the functional groups that are present in the given compound or the additional side chains connected at that position.
Where sec = 2 , Tert = 3
Complete Solution :
Given sec.butyl alcohol is the compound
In which the functional group is alcohol $\left( -OH \right)$ which is at the position of second carbon.
The structure of the compound is as follows:
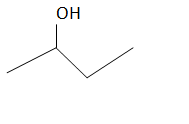
There are four carbons in this structure and alcohol functional group is at the second carbon which is having the IUPAC name as
2-butanol or butan-2-ol
Hence the option (B) is correct
- In option (A) the functional group is the position of first carbon which does not matches with the given compound hence it is not the correct option
- In option (C) the functional group is the position of first carbon and also an extra methyl group is added at the second which does not matches with the given compound hence it is not the correct option
- In option (D) the functional group is the position of second carbon and also an extra methyl group is added at the second which does not match with the given compound hence it is not the correct option.
So, the correct answer is “Option B”.
Note: It will be easy to write the IUPAC name if we draw the structure of the compound given. The full form of IUPAC is the International Union of Pure and Applied Chemistry. The IUPAC nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended by the International Union of Pure and Applied Chemistry. The names of the organic chemical compounds sometimes will write in simpler form as given in the question, that is sec. butyl alcohol.
Where sec = 2 , Tert = 3
Complete Solution :
Given sec.butyl alcohol is the compound
In which the functional group is alcohol $\left( -OH \right)$ which is at the position of second carbon.
The structure of the compound is as follows:

There are four carbons in this structure and alcohol functional group is at the second carbon which is having the IUPAC name as
2-butanol or butan-2-ol
Hence the option (B) is correct
- In option (A) the functional group is the position of first carbon which does not matches with the given compound hence it is not the correct option
- In option (C) the functional group is the position of first carbon and also an extra methyl group is added at the second which does not matches with the given compound hence it is not the correct option
- In option (D) the functional group is the position of second carbon and also an extra methyl group is added at the second which does not match with the given compound hence it is not the correct option.
So, the correct answer is “Option B”.
Note: It will be easy to write the IUPAC name if we draw the structure of the compound given. The full form of IUPAC is the International Union of Pure and Applied Chemistry. The IUPAC nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended by the International Union of Pure and Applied Chemistry. The names of the organic chemical compounds sometimes will write in simpler form as given in the question, that is sec. butyl alcohol.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




