The IUPAC name of ${\text{C}}{{\text{H}}_{\text{3}}}{\text{CH(C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{3}}}{\text{)C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{3}}}$ is
1.) 1, 1-methylethyl propane
2.) 2-ethylbutane
3.) 1-methyl-1-ethyl propane
4.) 3-methylpentane
Answer
621.9k+ views
Hint: The structure will be well understood when written in bond structural form. The carbon skeleton may or may not be a straight chain.
Complete step by step answer:
IUPAC is an abbreviation for International Union of Pure and Applied Chemistry.
IUPAC nomenclature is used for naming organic compound molecules. This method is used so that from the name we can construct an unmistakable structural formula. Inorganic chemistry also has an IUPAC nomenclature. Some compounds have long IUPAC names, in that case trivial names are used. While in some cases IUPAC names are simpler than trivial names.
Following the rules of IUPAC, we will see how to assign the nomenclature step by step.
Step 1:- The longest chain rule: The parent chain of the compound is the longest chain. It may be a straight chain or chain of other shapes. Following this rule, the parent chain of the compound given is as follows:
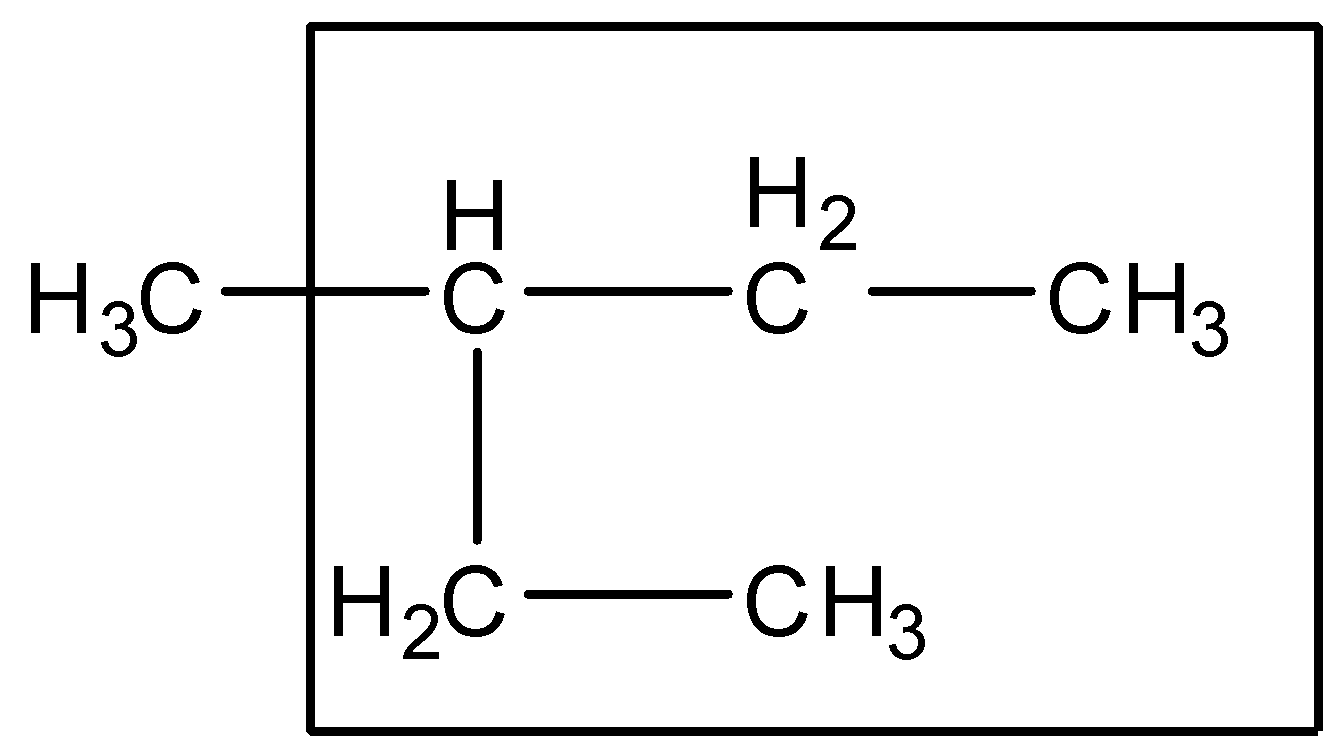
So accordingly we can see that the longest chain consists of 5 carbon atoms. Therefore the parent chain is Pentane.
Step 2:- The lowest set of locants: The carbons which belong to the longest carbon chain should be numbered. The numbers should begin from the end in which the lowest number is given to the carbon atom attached to the substituents.
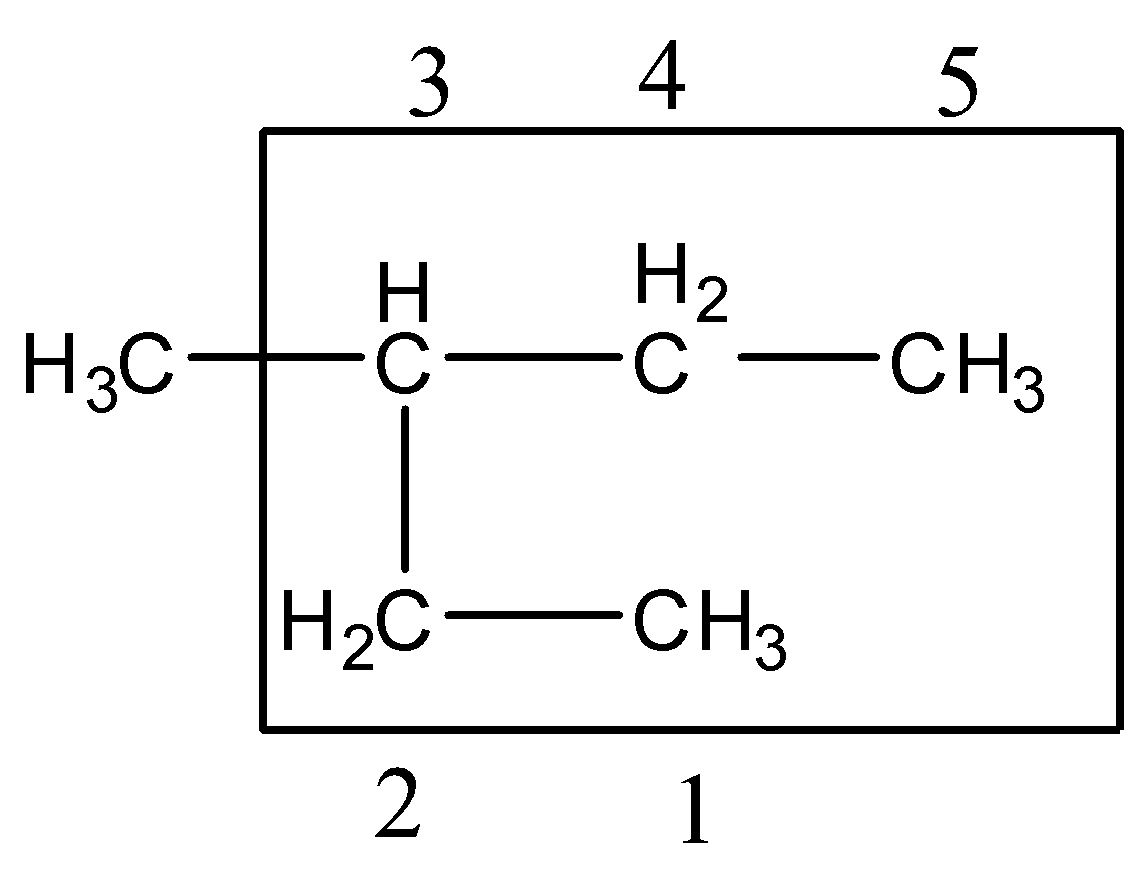
Since the substituent is attached to the center carbon atom, we can number from any one end. The substituent gets number 3.
Step 3:- Multiple instances of the same substituents: prefixes are used which indicate the total number of same substituents. Prefixes are di, tri, etc.
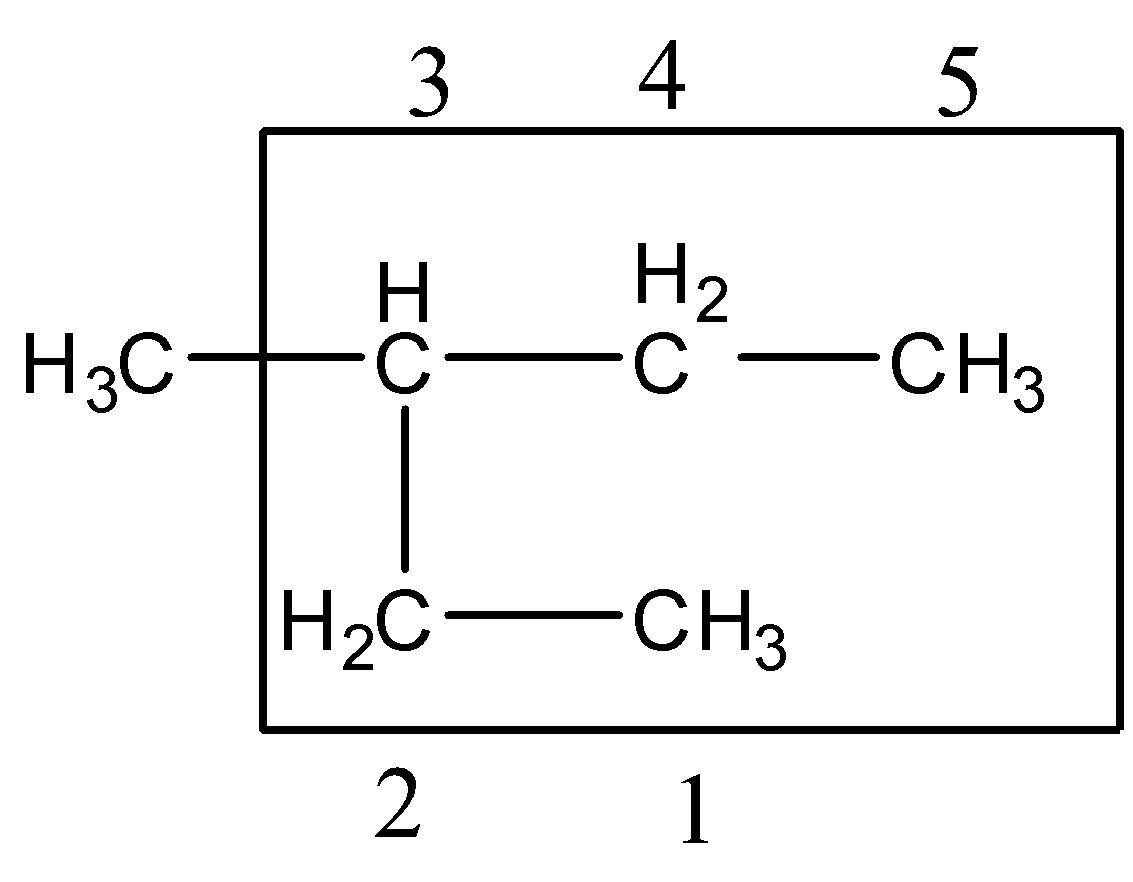
There is only one substitute attached so no need for any prefix.
The format of the IUPAC nomenclature is as follows: Locant + prefix + root + locanto +suffix.
As there is no functional group attached there is no need for the last two parts.
So the IUPAC name for ${\text{C}}{{\text{H}}_{\text{3}}}{\text{CH(C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{3}}}{\text{)C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{3}}}$ will be: 3-Methylpentane.
So, the correct answer is “Option D”.
Note: If there are different substituents attached to the carbon chain, they are arranged then in the alphabetical order of names. If there is any functional group attached to the carbon chain then they are written in the form of suffix.
Complete step by step answer:
IUPAC is an abbreviation for International Union of Pure and Applied Chemistry.
IUPAC nomenclature is used for naming organic compound molecules. This method is used so that from the name we can construct an unmistakable structural formula. Inorganic chemistry also has an IUPAC nomenclature. Some compounds have long IUPAC names, in that case trivial names are used. While in some cases IUPAC names are simpler than trivial names.
Following the rules of IUPAC, we will see how to assign the nomenclature step by step.
Step 1:- The longest chain rule: The parent chain of the compound is the longest chain. It may be a straight chain or chain of other shapes. Following this rule, the parent chain of the compound given is as follows:

So accordingly we can see that the longest chain consists of 5 carbon atoms. Therefore the parent chain is Pentane.
Step 2:- The lowest set of locants: The carbons which belong to the longest carbon chain should be numbered. The numbers should begin from the end in which the lowest number is given to the carbon atom attached to the substituents.

Since the substituent is attached to the center carbon atom, we can number from any one end. The substituent gets number 3.
Step 3:- Multiple instances of the same substituents: prefixes are used which indicate the total number of same substituents. Prefixes are di, tri, etc.

There is only one substitute attached so no need for any prefix.
The format of the IUPAC nomenclature is as follows: Locant + prefix + root + locanto +suffix.
As there is no functional group attached there is no need for the last two parts.
So the IUPAC name for ${\text{C}}{{\text{H}}_{\text{3}}}{\text{CH(C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{3}}}{\text{)C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{3}}}$ will be: 3-Methylpentane.
So, the correct answer is “Option D”.
Note: If there are different substituents attached to the carbon chain, they are arranged then in the alphabetical order of names. If there is any functional group attached to the carbon chain then they are written in the form of suffix.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




