The halo compound of methane used as fire extinguisher under the name pyrene is:
A. \[CHC{l_3}\]
B. \[C{H_2}C{l_2}\]
C. \[CC{l_2}{F_2}\]
D. \[CC{l_4}\]
Answer
607.8k+ views
Hint: In contradiction to the polycyclic aromatic hydrocarbon with the molecular formula \[{C_{16}}{H_{10}}\] , the Pyrene compound in consideration in this question is halo compound of methane, which is used as a fire extinguisher.
Complete Step-by-Step Solution:
Halo compounds of methane are basically the compounds in which one or more hydrogen atoms on the carbon atom in methane get replaced by a halogen.
Pyrene is a compound which has been used for a long time as a fire extinguisher. In 1910, a certain company in Delaware had patented the use of tetrachloromethane as a fire extinguishing compound. The compound was named after the company which patented it, i.e. Pyrene Manufacturing Company of Delaware. The heat of combustion caused the liquid to vaporize and this is what extinguished the flames. When carbon tetrachloride is heated to decomposition, it will emit fumes of toxic gases like phosgene and hydrogen chloride.
The chemical formula of pyrene is \[CC{l_4}\] and the molecular structure of the compound can be represented as follows:
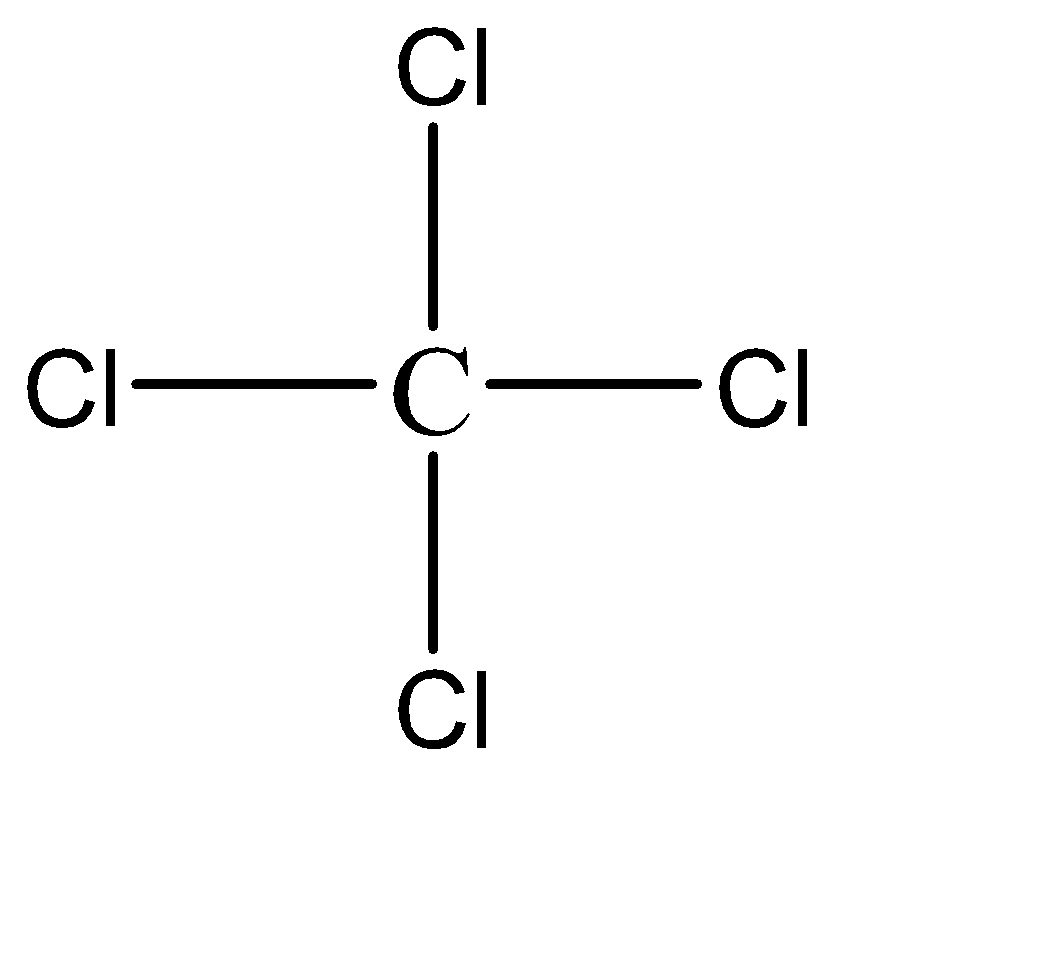
Hence, Option D is the correct option
Note: Some of the other common names used for pyrene are carbon tet (which is used in the cleaning industry), Halon (used in firefighting) and Refrigerant-10 (used in HVACR) carbon tetrachloride is used as a fire extinguisher because of two main reasons: it has no flash point and it is not flammable
Complete Step-by-Step Solution:
Halo compounds of methane are basically the compounds in which one or more hydrogen atoms on the carbon atom in methane get replaced by a halogen.
Pyrene is a compound which has been used for a long time as a fire extinguisher. In 1910, a certain company in Delaware had patented the use of tetrachloromethane as a fire extinguishing compound. The compound was named after the company which patented it, i.e. Pyrene Manufacturing Company of Delaware. The heat of combustion caused the liquid to vaporize and this is what extinguished the flames. When carbon tetrachloride is heated to decomposition, it will emit fumes of toxic gases like phosgene and hydrogen chloride.
The chemical formula of pyrene is \[CC{l_4}\] and the molecular structure of the compound can be represented as follows:

Hence, Option D is the correct option
Note: Some of the other common names used for pyrene are carbon tet (which is used in the cleaning industry), Halon (used in firefighting) and Refrigerant-10 (used in HVACR) carbon tetrachloride is used as a fire extinguisher because of two main reasons: it has no flash point and it is not flammable
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




