The geometry of methane molecule is:
(A) Tetrahedral
(B) Pyramidal
(C) Octahedral
(D) Square planar
Answer
550.5k+ views
Hint :To answer this question, we need to understand the concept of geometry of carbon-based organic compounds. Geometry is based on the concept of hybridization of atoms.
Complete Step By Step Answer:
Methane is a saturated hydrocarbon and it contains only single bonds. A molecule of methane has a single carbon atom and four hydrogen atoms. The geometry of the methane molecule is tetrahedral. It exhibits sp3 hybridization where a carbon atom is at the center and four hydrogen atoms are at the four corners. The bond length is about 109 pm and the angle between two hydrogen atoms in the arrangement is about 109.5 degrees. The methane molecule has a molecular weight of 16.04.
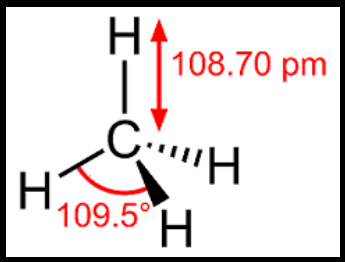
Hence, the correct option is (a).
Note :
Methane is a principal component of natural gas. When a single molecule of methane is burned in the presence of oxygen it releases one molecule of $CO_2$ (carbon dioxide) and two molecules of $H_2O$ (water). The strength of the carbon hydrogen covalent bond in methane is among the strongest in all hydrocarbons. Pure methane is odorless, but when used as a fuel it is usually mixed with small quantities of strongly-smelling sulfur compounds such as ethyl mercaptan to enable the detection of leaks.
Complete Step By Step Answer:
Methane is a saturated hydrocarbon and it contains only single bonds. A molecule of methane has a single carbon atom and four hydrogen atoms. The geometry of the methane molecule is tetrahedral. It exhibits sp3 hybridization where a carbon atom is at the center and four hydrogen atoms are at the four corners. The bond length is about 109 pm and the angle between two hydrogen atoms in the arrangement is about 109.5 degrees. The methane molecule has a molecular weight of 16.04.

Hence, the correct option is (a).
Note :
Methane is a principal component of natural gas. When a single molecule of methane is burned in the presence of oxygen it releases one molecule of $CO_2$ (carbon dioxide) and two molecules of $H_2O$ (water). The strength of the carbon hydrogen covalent bond in methane is among the strongest in all hydrocarbons. Pure methane is odorless, but when used as a fuel it is usually mixed with small quantities of strongly-smelling sulfur compounds such as ethyl mercaptan to enable the detection of leaks.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




