The enthalpy of hydrogenation of cyclohexene is $ - 119.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$. If resonance energy of benzene is $ - 150.4{\text{ kJ mo}}{{\text{l}}^{ - 1}}$, its enthalpy of hydrogenation would be
A) $ - 508.9{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
B) $ - 208.1{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
C) $ - 269.9{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
D) $ - 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
Answer
598.2k+ views
Hint: We know that benzene $\left( {{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}\left( {\text{l}} \right)} \right)$ contains three double bonds. To break these three bonds and convert benzene $\left( {{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}\left( {\text{l}} \right)} \right)$ to cyclohexane $\left( {{{\text{C}}_{\text{6}}}{{\text{H}}_{{\text{12}}}}\left( {\text{l}} \right)} \right)$ three molecules of hydrogen $\left( {{{\text{H}}_{\text{2}}}} \right)$ will be required. To calculate the enthalpy of hydrogenation of the given compound we must add resonance energy to the total enthalpy of the reaction.
Complete solution:
We are given that the enthalpy of hydrogenation of cyclohexene is $ - 119.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$. The enthalpy of hydrogenation of cyclohexene is the enthalpy for addition of one molecule of hydrogen to cyclohexane.
The enthalpy of hydrogenation of benzene is the enthalpy for addition of one molecule of hydrogen to cyclohexane. The hydrogenation reaction of benzene is as follows:
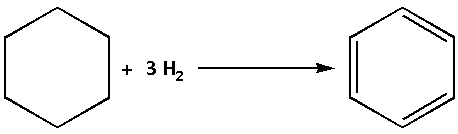
We are given that the enthalpy of hydrogenation of cyclohexene is $ - 119.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$. Thus, the enthalpy for hydrogenation of benzene is,
$\Delta {{\text{H}}_{{\text{benzene}}}} = 3 \times \Delta {{\text{H}}_{{\text{cyclohexene}}}}$
Where $\Delta {{\text{H}}_{{\text{cyclohexene}}}}$ is the enthalpy of hydrogenation of cyclohexene,
$\Delta {{\text{H}}_{{\text{benzene}}}}$ is the enthalpy of hydrogenation of benzene.
$\Delta {{\text{H}}_{{\text{benzene}}}} = 3 \times - 119.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
$\Delta {{\text{H}}_{{\text{benzene}}}} = - 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
Thus, the enthalpy of hydrogenation of benzene is $ - 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$.
We know that benzene is a resonating structure. Thus, benzene possesses resonance energy. The amount of energy required to convert the true delocalized structure into stable structure is known as resonating energy. Thus,
${\text{Resonance energy}} = \Delta {{\text{H}}_{{\text{benzene}}}} - \Delta {{\text{H}}_{{\text{benzene (actual)}}}}$
We are given that the resonance energy of benzene is $ - 150.4{\text{ kJ mo}}{{\text{l}}^{ - 1}}$. Thus,
$ - 150.4{\text{ kJ mo}}{{\text{l}}^{ - 1}} = - 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}} - \Delta {{\text{H}}_{{\text{benzene (actual)}}}}$
$ - \Delta {{\text{H}}_{{\text{benzene (actual)}}}} = - 150.4{\text{ kJ mo}}{{\text{l}}^{ - 1}} + 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
$ - \Delta {{\text{H}}_{{\text{benzene (actual)}}}} = 208.1{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
$\Delta {{\text{H}}_{{\text{benzene (actual)}}}} = - 208.1{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
Thus, the enthalpy of hydrogenation of benzene is $ - 208.1{\text{ kJ mo}}{{\text{l}}^{ - 1}}$.
Thus, the correct option is (C) 8.87.
Note:The amount of energy required to convert the true delocalized structure into stable structure is known as resonating energy. The resonance energy is the measure of extra stability of a conjugated system compared to the number of isolated double bonds.
Complete solution:
We are given that the enthalpy of hydrogenation of cyclohexene is $ - 119.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$. The enthalpy of hydrogenation of cyclohexene is the enthalpy for addition of one molecule of hydrogen to cyclohexane.
The enthalpy of hydrogenation of benzene is the enthalpy for addition of one molecule of hydrogen to cyclohexane. The hydrogenation reaction of benzene is as follows:
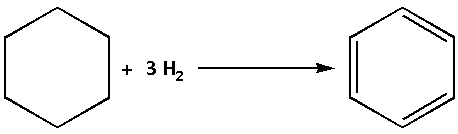
We are given that the enthalpy of hydrogenation of cyclohexene is $ - 119.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$. Thus, the enthalpy for hydrogenation of benzene is,
$\Delta {{\text{H}}_{{\text{benzene}}}} = 3 \times \Delta {{\text{H}}_{{\text{cyclohexene}}}}$
Where $\Delta {{\text{H}}_{{\text{cyclohexene}}}}$ is the enthalpy of hydrogenation of cyclohexene,
$\Delta {{\text{H}}_{{\text{benzene}}}}$ is the enthalpy of hydrogenation of benzene.
$\Delta {{\text{H}}_{{\text{benzene}}}} = 3 \times - 119.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
$\Delta {{\text{H}}_{{\text{benzene}}}} = - 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
Thus, the enthalpy of hydrogenation of benzene is $ - 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$.
We know that benzene is a resonating structure. Thus, benzene possesses resonance energy. The amount of energy required to convert the true delocalized structure into stable structure is known as resonating energy. Thus,
${\text{Resonance energy}} = \Delta {{\text{H}}_{{\text{benzene}}}} - \Delta {{\text{H}}_{{\text{benzene (actual)}}}}$
We are given that the resonance energy of benzene is $ - 150.4{\text{ kJ mo}}{{\text{l}}^{ - 1}}$. Thus,
$ - 150.4{\text{ kJ mo}}{{\text{l}}^{ - 1}} = - 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}} - \Delta {{\text{H}}_{{\text{benzene (actual)}}}}$
$ - \Delta {{\text{H}}_{{\text{benzene (actual)}}}} = - 150.4{\text{ kJ mo}}{{\text{l}}^{ - 1}} + 358.5{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
$ - \Delta {{\text{H}}_{{\text{benzene (actual)}}}} = 208.1{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
$\Delta {{\text{H}}_{{\text{benzene (actual)}}}} = - 208.1{\text{ kJ mo}}{{\text{l}}^{ - 1}}$
Thus, the enthalpy of hydrogenation of benzene is $ - 208.1{\text{ kJ mo}}{{\text{l}}^{ - 1}}$.
Thus, the correct option is (C) 8.87.
Note:The amount of energy required to convert the true delocalized structure into stable structure is known as resonating energy. The resonance energy is the measure of extra stability of a conjugated system compared to the number of isolated double bonds.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




