The electronic configuration in which Pauli’s exclusion principle or Hund’s rule is not violated is _______
A.

B.
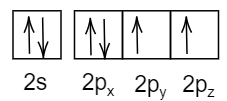
C.
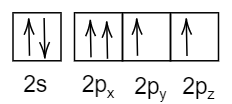
D.




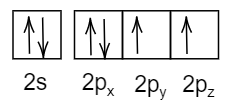
Answer
601.8k+ views
Hint: Remember that, the distribution of electrons in different orbitals is termed as electronic configuration. Three rules should be followed while deciding the electronic configuration of an atom and they are Hund’s rule, Pauli’s exclusion principle and Aufbau’s principle.
Complete Solution :
Let’s discuss Pauli’s exclusion principle, all subshells in an orbital can accommodate a maximum of two electrons and spin of both the electrons is opposite to each other.
While, according to Hund’s rule each subshell in an orbital must be filled with one electron each before anyone is doubly occupied and the spin of all electrons in singly occupied shells is the same.
Now, let us identify the correct answer from the options given in the question.
- In first option:

The configuration is violating Hund’s rule where the last subshell of p-orbital should be filled first before doubly occupying the first orbital of p-orbital.
- In second option:
The electronic configuration follows both Hund’s rule and Pauli’s exclusion principle.
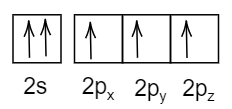
- In the third option:
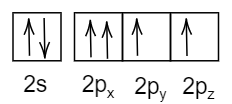
In this respective electronic configuration, it violates Pauli’s exclusion principle where the spins in the p-orbital should be opposite in direction, not in the same direction.
- In the last option:
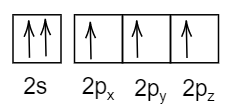
All the spins are in the same direction and thus it violates Pauli's exclusion principle.
Therefore, we can conclude the only option that does not violate Hund’s rule and Pauli’s exclusion principle is the second option.
So, the correct answer is “Option B”.
Note: Aufbau principle should be followed while drawing an electronic configuration of an atom. According to this principle, electrons should be filled in lower energy orbitals first and then must be filled in higher energy orbitals.
Complete Solution :
Let’s discuss Pauli’s exclusion principle, all subshells in an orbital can accommodate a maximum of two electrons and spin of both the electrons is opposite to each other.
While, according to Hund’s rule each subshell in an orbital must be filled with one electron each before anyone is doubly occupied and the spin of all electrons in singly occupied shells is the same.
Now, let us identify the correct answer from the options given in the question.
- In first option:

The configuration is violating Hund’s rule where the last subshell of p-orbital should be filled first before doubly occupying the first orbital of p-orbital.
- In second option:

The electronic configuration follows both Hund’s rule and Pauli’s exclusion principle.
- In the third option:

In this respective electronic configuration, it violates Pauli’s exclusion principle where the spins in the p-orbital should be opposite in direction, not in the same direction.
- In the last option:

All the spins are in the same direction and thus it violates Pauli's exclusion principle.
Therefore, we can conclude the only option that does not violate Hund’s rule and Pauli’s exclusion principle is the second option.
So, the correct answer is “Option B”.
Note: Aufbau principle should be followed while drawing an electronic configuration of an atom. According to this principle, electrons should be filled in lower energy orbitals first and then must be filled in higher energy orbitals.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




