The correct IUPAC name is:
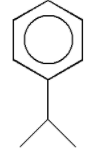
A) Isopropyl benzene
B) Cumene
C) Phenylisopropyl
D) None of these

Answer
603k+ views
Hint:The method of naming organic compounds is known as the IUPAC (International Union for Pure and Applied Chemistry) nomenclature. IUPAC is a common naming system which enlists standard rules. In the given structure, a benzene ring is present. An isopropyl group is attached to the benzene ring.
Complete answer:
The given structure with all of its atoms is as follows:
We know that the given structure contains a benzene ring. Thus, it is an aromatic compound. The group ${{\text{H}}_{\text{3}}}{\text{C}} - {\text{CH}} - {\text{C}}{{\text{H}}_{\text{3}}}$ is the substituent which is aliphatic in nature. Thus, in the given structure an aliphatic substituent is attached to an aromatic ring.
The group ${{\text{H}}_{\text{3}}}{\text{C}} - {\text{CH}} - {\text{C}}{{\text{H}}_{\text{3}}}$ contains three carbon atoms and one hydrogen atom less than propane. Thus, the group ${{\text{H}}_{\text{3}}}{\text{C}} - {\text{CH}} - {\text{C}}{{\text{H}}_{\text{3}}}$ is known as an isopropyl group. In the given structure, an isopropyl group is attached to the benzene ring.
According to IUPAC nomenclature, while naming the aromatic compounds, we first write the name of the substituent group as a prefix. Thus, the name of the given compound is isopropyl benzene. Thus, the correct IUPAC name is isopropyl benzene.
Additional Information
Isopropyl benzene is an organic compound. It is an aromatic hydrocarbon with an aliphatic substitution. Isopropyl benzene is colourless liquid in appearance. It is highly flammable in nature.
Thus, the correct answer is option (A) isopropyl benzene.
Note:The common name of the given compound is cumene. But we are asked to write the IUPAC name of the compound which is isopropyl benzene and not cumene. Do not confuse between the common name and IUPAC name. Other names for the given compound are (propan-2-yl)benzene, (1-methylethyl)benzene, and cumol.
Complete answer:
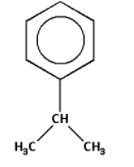
The given structure with all of its atoms is as follows:

We know that the given structure contains a benzene ring. Thus, it is an aromatic compound. The group ${{\text{H}}_{\text{3}}}{\text{C}} - {\text{CH}} - {\text{C}}{{\text{H}}_{\text{3}}}$ is the substituent which is aliphatic in nature. Thus, in the given structure an aliphatic substituent is attached to an aromatic ring.
The group ${{\text{H}}_{\text{3}}}{\text{C}} - {\text{CH}} - {\text{C}}{{\text{H}}_{\text{3}}}$ contains three carbon atoms and one hydrogen atom less than propane. Thus, the group ${{\text{H}}_{\text{3}}}{\text{C}} - {\text{CH}} - {\text{C}}{{\text{H}}_{\text{3}}}$ is known as an isopropyl group. In the given structure, an isopropyl group is attached to the benzene ring.
According to IUPAC nomenclature, while naming the aromatic compounds, we first write the name of the substituent group as a prefix. Thus, the name of the given compound is isopropyl benzene. Thus, the correct IUPAC name is isopropyl benzene.
Additional Information
Isopropyl benzene is an organic compound. It is an aromatic hydrocarbon with an aliphatic substitution. Isopropyl benzene is colourless liquid in appearance. It is highly flammable in nature.
Thus, the correct answer is option (A) isopropyl benzene.
Note:The common name of the given compound is cumene. But we are asked to write the IUPAC name of the compound which is isopropyl benzene and not cumene. Do not confuse between the common name and IUPAC name. Other names for the given compound are (propan-2-yl)benzene, (1-methylethyl)benzene, and cumol.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




