The compound that is most reactive towards electrophilic nitration is:
A. Toluene
B. Benzene
C. Benzoic acid
D. Nitrobenzene
Answer
585.6k+ views
Hint: We know that nitration is a reaction in which a nitro group displaces a hydrogen atom in a compound. A nitro group is an electron withdrawing group and nitration happens to that compound if the compound has an electron releasing group.
Complete step by step answer:
Let’s discuss the possibility of electrophilic nitration in all cases.
Option A is toluene. The structure of toluene is,
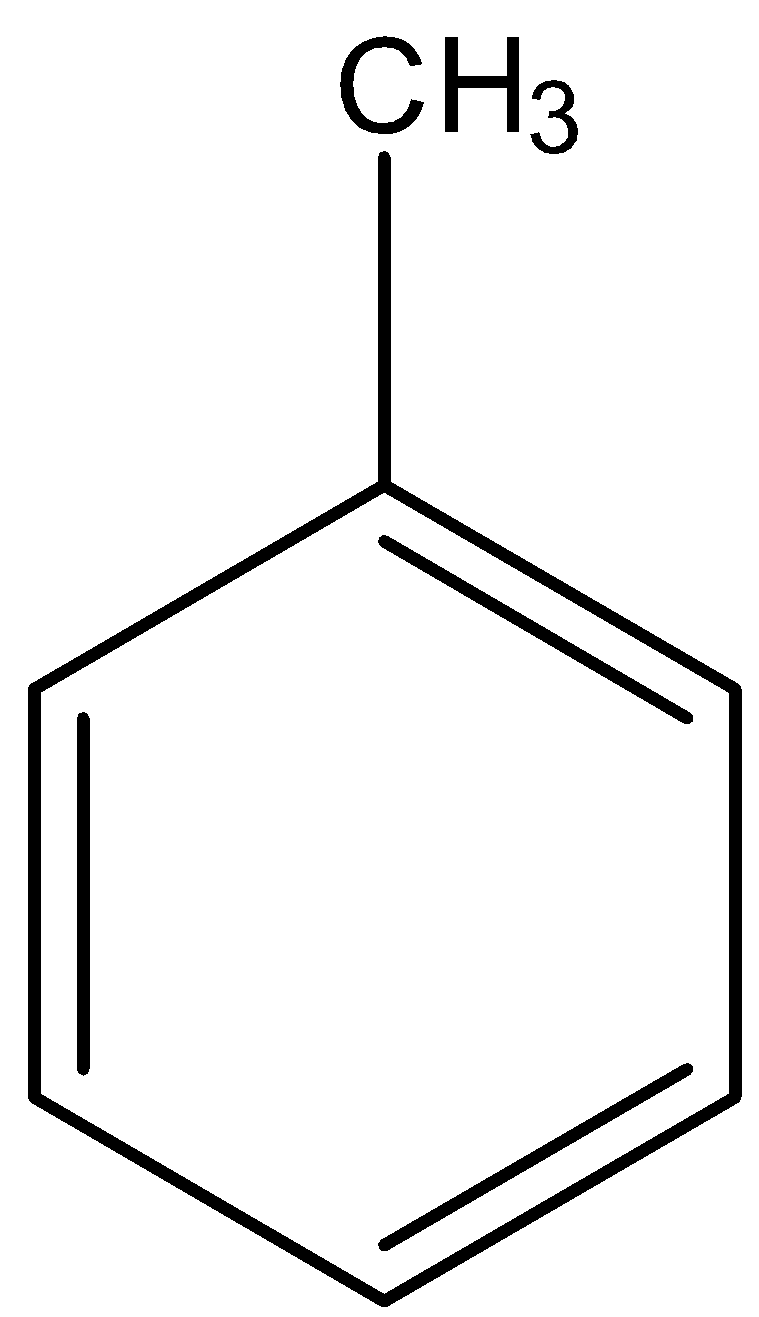
We know that the $CH_3$ group is an electron releasing group. And we know that the nitro group is an electron withdrawing group. So, the electrophilic substitution of the $NO_2$ group is possible here.
Option B is benzene. The structure of benzene is,
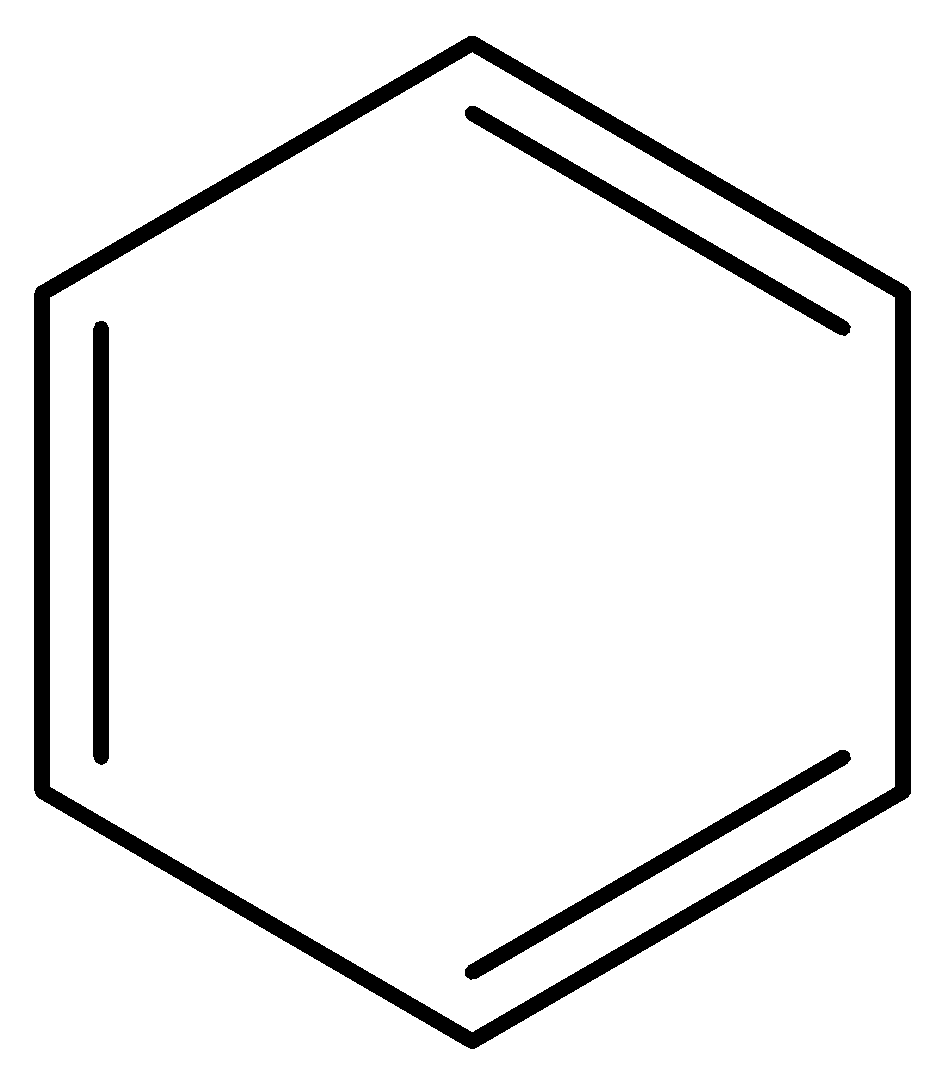
Here, also nitration is possible. But its reactivity towards electrophilic nitration is less than toluene because of absence of electron releasing group. Option C is benzoic acid. The structure of benzoic acid is,
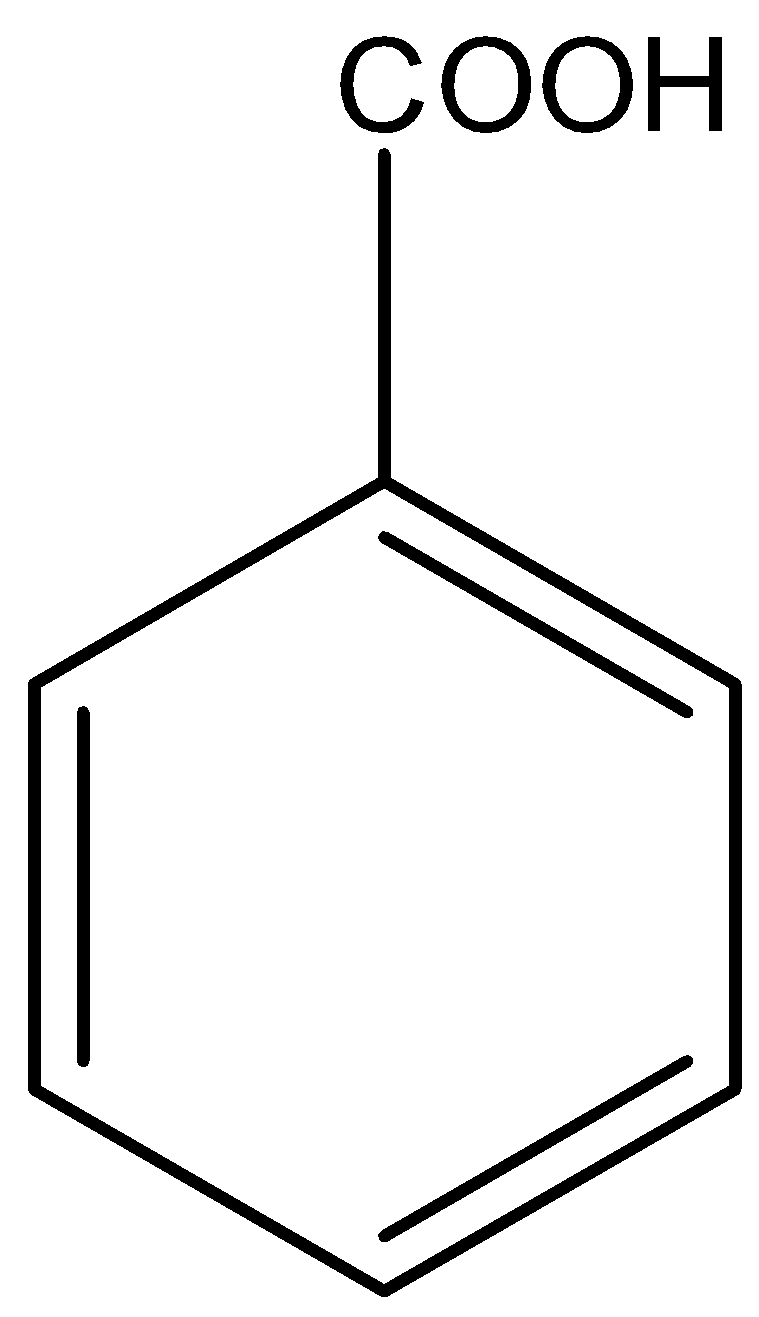
Here, COOH group is an electron withdrawing group. So, electrophilic nitration is less reactive in benzoic acid than benzene as the nitro group is also an electron withdrawing group.
Option D is nitrobenzene. The structure of nitrobenzene is,
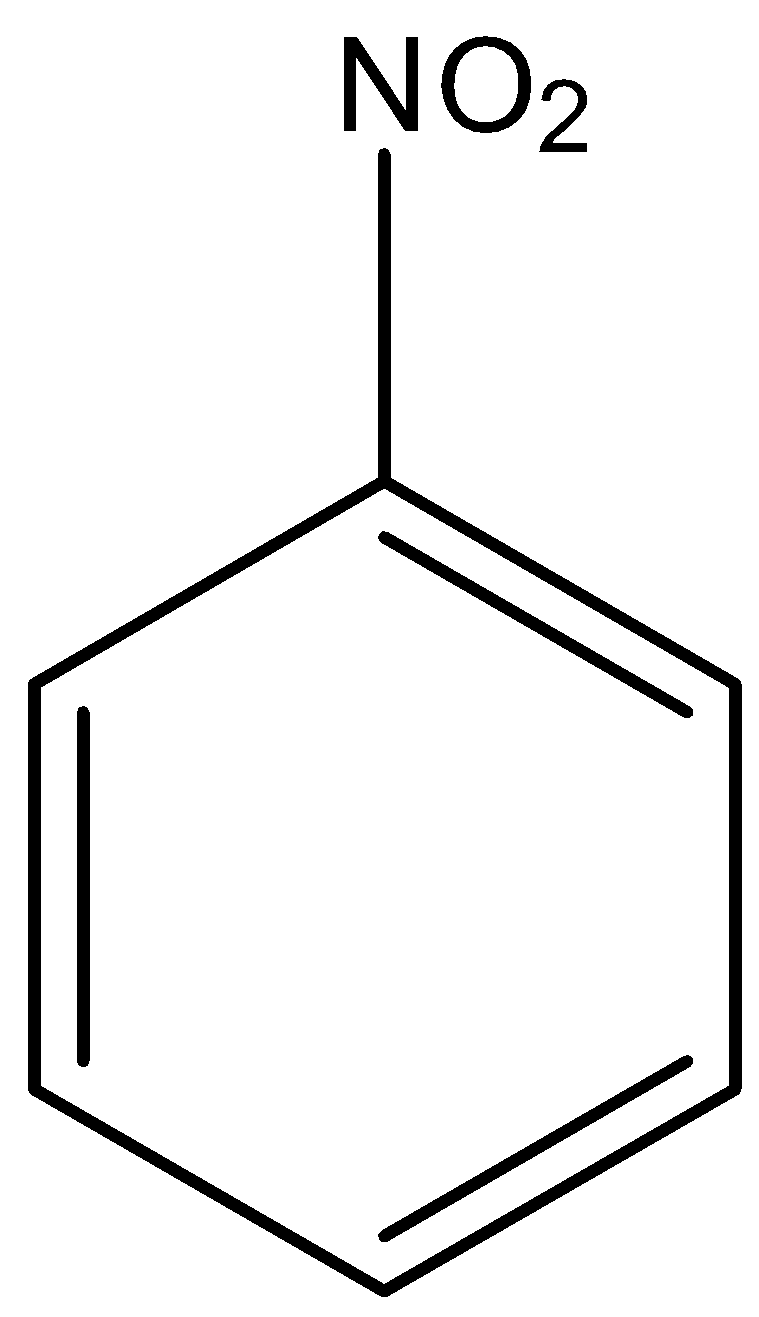
We know that the nitro group is an electron withdrawing group. So, electrophilic nitration is less reactive than benzene.
So, the compound which is more reactive towards electrophilic nitration is toluene.
So, the correct answer is Option A.
Note: Always remember that substituents having pi bonds to electronegative atoms (C=O, -$NO_2$) adjacent to the pi system are electron withdrawing groups. They deactivate the aromatic ring by decreasing electron density on the ring by resonance.
Complete step by step answer:
Let’s discuss the possibility of electrophilic nitration in all cases.
Option A is toluene. The structure of toluene is,

We know that the $CH_3$ group is an electron releasing group. And we know that the nitro group is an electron withdrawing group. So, the electrophilic substitution of the $NO_2$ group is possible here.
Option B is benzene. The structure of benzene is,

Here, also nitration is possible. But its reactivity towards electrophilic nitration is less than toluene because of absence of electron releasing group. Option C is benzoic acid. The structure of benzoic acid is,

Here, COOH group is an electron withdrawing group. So, electrophilic nitration is less reactive in benzoic acid than benzene as the nitro group is also an electron withdrawing group.
Option D is nitrobenzene. The structure of nitrobenzene is,

We know that the nitro group is an electron withdrawing group. So, electrophilic nitration is less reactive than benzene.
So, the compound which is more reactive towards electrophilic nitration is toluene.
So, the correct answer is Option A.
Note: Always remember that substituents having pi bonds to electronegative atoms (C=O, -$NO_2$) adjacent to the pi system are electron withdrawing groups. They deactivate the aromatic ring by decreasing electron density on the ring by resonance.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




