The compound having only primary hydrogen atoms is:
A. isobutene
B. \[2,3 - \]Dimethyl\[ - 1 - \]butene
C. cyclohexane
D. propane
Answer
576.3k+ views
Hint: To answer this question we should know what primary hydrogen is. The hydrogen attached with primary carbon is known as primary hydrogen. We will draw the geometry of all the given molecules then we will find the primary carbon to determine the compound having all primary hydrogen atoms.
Complete step-by-step answer:
A carbon atom has four valencies. Primary carbon is the carbon which is attached with one other carbon only other three valencies are satisfied by hydrogen atoms.
The primary carbon is shown as follows:
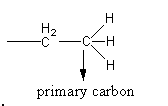
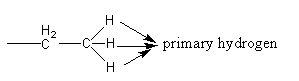
The hydrogen atoms attached with primary carbon are known as primary hydrogen atoms which are shown as follows:
The structure of the given compounds are as follows:
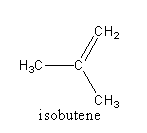
In isobutene, three carbon are primary because they are attached with only one carbon atom. So, all hydrogen is primary.
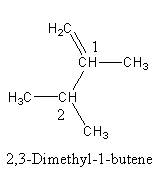
In \[2,3 - \]Dimethyl\[ - 1 - \]butene, four carbon are primary except the carbon indicated by $1$ and $2$. The carbon indicated by $1$ and $2$ also has one hydrogen atom.
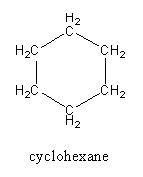
In cyclohexane, every carbon is attached with two other carbon atoms, so cyclohexane does not have any primary carbon hence no primary hydrogen atoms.
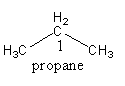
In propane, two primary carbon are present except the carbon indicated by $1$. The carbon indicated by $1$ also has two hydrogen atoms.
So, the compound having only primary hydrogen atoms is isobutene.
Therefore, option (A) isobutene, is correct.
Note: The carbon attached with two other carbon is known as secondary carbon and the hydrogen atoms attached with secondary carbon are known as secondary hydrogen atoms. The carbon attached with three other carbon is known as tertiary carbon and the hydrogen atoms attached with tertiary carbon are known as tertiary hydrogen atoms. Here, except cyclohexane, all have primary carbon and primary hydrogen but only isobutene has all primary hydrogen. Isobutene has tertiary carbon but no the tertiary hydrogen.
Complete step-by-step answer:
A carbon atom has four valencies. Primary carbon is the carbon which is attached with one other carbon only other three valencies are satisfied by hydrogen atoms.
The primary carbon is shown as follows:

The hydrogen atoms attached with primary carbon are known as primary hydrogen atoms which are shown as follows:

The structure of the given compounds are as follows:

In isobutene, three carbon are primary because they are attached with only one carbon atom. So, all hydrogen is primary.

In \[2,3 - \]Dimethyl\[ - 1 - \]butene, four carbon are primary except the carbon indicated by $1$ and $2$. The carbon indicated by $1$ and $2$ also has one hydrogen atom.

In cyclohexane, every carbon is attached with two other carbon atoms, so cyclohexane does not have any primary carbon hence no primary hydrogen atoms.

In propane, two primary carbon are present except the carbon indicated by $1$. The carbon indicated by $1$ also has two hydrogen atoms.
So, the compound having only primary hydrogen atoms is isobutene.
Therefore, option (A) isobutene, is correct.
Note: The carbon attached with two other carbon is known as secondary carbon and the hydrogen atoms attached with secondary carbon are known as secondary hydrogen atoms. The carbon attached with three other carbon is known as tertiary carbon and the hydrogen atoms attached with tertiary carbon are known as tertiary hydrogen atoms. Here, except cyclohexane, all have primary carbon and primary hydrogen but only isobutene has all primary hydrogen. Isobutene has tertiary carbon but no the tertiary hydrogen.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




