The colour of methyl orange indicator in a solution is yellow. The pH of this solution is likely to be:
A. 7
B. less than 7
C. 0
D. more than 7
Answer
598.8k+ views
Hint: When measuring pH, the negative logarithm of the concentration of hydrogen is taken. It can be only done if the hydrogen ions are able to move about freely in the solution, it only happens in a specific type of solution.
Complete answer:
In chemistry, pH is referred to as the potential of hydrogen. pH is a standard scale that is used to measure how much acidic or basic a solution is. pH was invented by S.P.L Sorensen and the scale ranges from the value of 0 to 14. As we progress right in the scale, the acidic character reduces and the basic character increases. Solutions which have pH close less than 7 are said to be acidic, whereas those with pH more than 7 are basic.
At pH=7 the solution is said to be neutral, as it has equal amounts of ${{H}^{+}}$ and $O{{H}^{-}}$ ions.
Here is the pH scale:
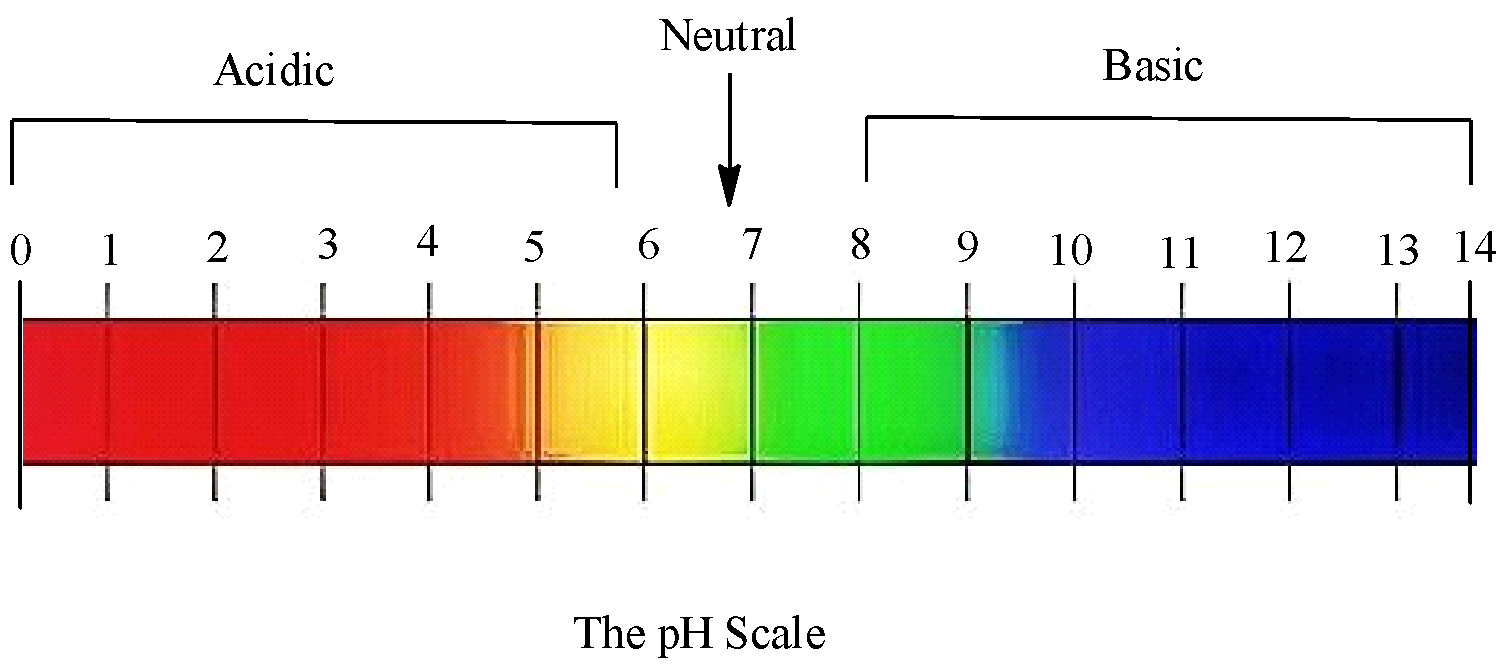
So, on dipping a pH paper in a solution, we get a colour and by identifying the colour, we can get the respective pH values. Mathematically pH is represented by: $pH=-\log [{{H}^{+}}]$, so pH depends on the concentration of hydrogen ions present in the solution. However, for that to happen the solution must be aqueous. Only in aqueous solutions the ions get dissociated, and dissociation does not take place in non-aqueous solutions.
Now ,in the case of methyl orange indicator, it turns yellow when a basic or alkaline substance is used.
Hence the pH of the solution is more than 7 in this case. So, our correct answer would be option D.
NOTE: As $pH=-\log [{{H}^{+}}]$, hence it is a decreasing function. A solution that has more ${{H}^{+}}$ in it will have more pH. Moreover, the pH of pure water at ${{25}^{0}}C$ is equal to 7. Temperature also plays a role in determining the pH.
Complete answer:
In chemistry, pH is referred to as the potential of hydrogen. pH is a standard scale that is used to measure how much acidic or basic a solution is. pH was invented by S.P.L Sorensen and the scale ranges from the value of 0 to 14. As we progress right in the scale, the acidic character reduces and the basic character increases. Solutions which have pH close less than 7 are said to be acidic, whereas those with pH more than 7 are basic.
At pH=7 the solution is said to be neutral, as it has equal amounts of ${{H}^{+}}$ and $O{{H}^{-}}$ ions.
Here is the pH scale:

So, on dipping a pH paper in a solution, we get a colour and by identifying the colour, we can get the respective pH values. Mathematically pH is represented by: $pH=-\log [{{H}^{+}}]$, so pH depends on the concentration of hydrogen ions present in the solution. However, for that to happen the solution must be aqueous. Only in aqueous solutions the ions get dissociated, and dissociation does not take place in non-aqueous solutions.
Now ,in the case of methyl orange indicator, it turns yellow when a basic or alkaline substance is used.
Hence the pH of the solution is more than 7 in this case. So, our correct answer would be option D.
NOTE: As $pH=-\log [{{H}^{+}}]$, hence it is a decreasing function. A solution that has more ${{H}^{+}}$ in it will have more pH. Moreover, the pH of pure water at ${{25}^{0}}C$ is equal to 7. Temperature also plays a role in determining the pH.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE

10 examples of friction in our daily life




