
The cationic part of the solid $Xe{{F}_{6}}$ is having the ‘______’ shape.
(a)- Linear
(b)- Angular
(c)- Square pyramidal
(d)- Tetrahedral
Answer
592.2k+ views
Hint: The cationic part of $Xe{{F}_{6}}$ is based on the number of atoms joined to the central atom. Not only atoms, but lone pairs are also considered for the shape. For finding the shape of the compound the number of the total number of electron pairs and the number of lone pairs should be calculated with the help of valence electrons, the number of bonds, etc.
Complete answer:
The reaction of $Xe{{F}_{6}}$ to form cation and anion is:
$Xe{{F}_{6}}\to Xe{{F}_{5}}^{+}+{{F}^{-}}$
So, we have to find the structure of $Xe{{F}_{5}}^{+}$.
Both the VSEPR theory and the concept of hybridization are applied to predict the molecular geometries of xenon compounds.
According to the VSEPR theory, the shape of the molecule is predicted by the total number of electron pairs (lone pairs + bond pairs) in the valence shell of the central Xe atom.
To calculate the total number of electron pairs:
\[\dfrac{\text{valence electrons of central atom + number of bonded atoms}}{\text{2}}\]
Since 5 fluorine atoms are bonded to the xenon atom and the molecule has a positive charge hence one electron will be reduced.
With the above formula: \[\dfrac{8+(5-1)}{2}=\dfrac{8+4}{2}=6\]
Hence, there are 6 electron pairs.
Since there are 5 fluorine atoms attached to xenon, there are 5 bond pairs of electrons.
Now for calculating the number of lone pairs in the compound: -
Total number of electron pairs –number of bond pairs.
Lone pairs = 6 – 5 = 1
Hence, in the compound, there are 1 lone pair.
Depending on the number of \[Xe-F\] covalent bonds to be formed, the requisite number of electrons of the \[5p-orbital\] valence shell of Xe gets unpaired and promoted to the vacant \[5d-orbitals\] followed by hybridization.
Since there are 5 bond pairs of an electron and one lone pair hence the hybridization is $s{{p}^{3}}{{d}^{2}}$.
So, the hybridization is $s{{p}^{3}}{{d}^{2}}$ and has one lone pair the structure of $Xe{{F}_{5}}^{+}$ is square pyramidal.
The structure is given below:
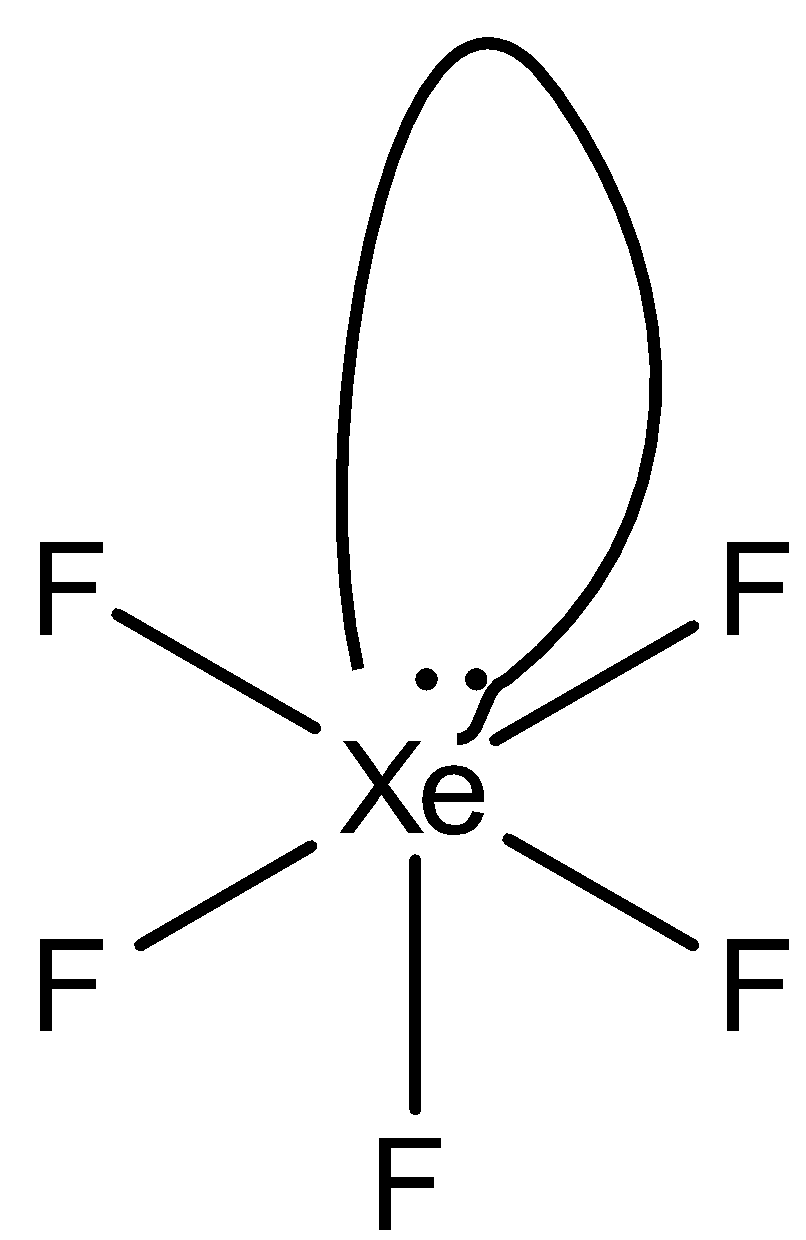
So, the correct answer is “Option C”.
Note: Whenever you are drawing the compound structure the number of lone pairs should also be considered. For example, when 4 atoms of fluorine are bonded with xenon you could get confused between tetrahedral, square planar shape, and Trigonal bipyramidal.
Complete answer:
The reaction of $Xe{{F}_{6}}$ to form cation and anion is:
$Xe{{F}_{6}}\to Xe{{F}_{5}}^{+}+{{F}^{-}}$
So, we have to find the structure of $Xe{{F}_{5}}^{+}$.
Both the VSEPR theory and the concept of hybridization are applied to predict the molecular geometries of xenon compounds.
According to the VSEPR theory, the shape of the molecule is predicted by the total number of electron pairs (lone pairs + bond pairs) in the valence shell of the central Xe atom.
To calculate the total number of electron pairs:
\[\dfrac{\text{valence electrons of central atom + number of bonded atoms}}{\text{2}}\]
Since 5 fluorine atoms are bonded to the xenon atom and the molecule has a positive charge hence one electron will be reduced.
With the above formula: \[\dfrac{8+(5-1)}{2}=\dfrac{8+4}{2}=6\]
Hence, there are 6 electron pairs.
Since there are 5 fluorine atoms attached to xenon, there are 5 bond pairs of electrons.
Now for calculating the number of lone pairs in the compound: -
Total number of electron pairs –number of bond pairs.
Lone pairs = 6 – 5 = 1
Hence, in the compound, there are 1 lone pair.
Depending on the number of \[Xe-F\] covalent bonds to be formed, the requisite number of electrons of the \[5p-orbital\] valence shell of Xe gets unpaired and promoted to the vacant \[5d-orbitals\] followed by hybridization.
Since there are 5 bond pairs of an electron and one lone pair hence the hybridization is $s{{p}^{3}}{{d}^{2}}$.
So, the hybridization is $s{{p}^{3}}{{d}^{2}}$ and has one lone pair the structure of $Xe{{F}_{5}}^{+}$ is square pyramidal.
The structure is given below:

So, the correct answer is “Option C”.
Note: Whenever you are drawing the compound structure the number of lone pairs should also be considered. For example, when 4 atoms of fluorine are bonded with xenon you could get confused between tetrahedral, square planar shape, and Trigonal bipyramidal.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




