The atomicity of phosphorus is X and the P-P-P bond angle is Y. What are X and Y?
Answer
617.7k+ views
Hint: To solve this question, we need to know the regular tetrahedron structure of phosphorus. As we see in the figure below, we have 6 P-P single bonds and also 4 lone pairs of electrons in this molecule. The bond length of P-P is 2.21 A and P-P-P angle and atomicity is asked in this question.
Complete step by step solution:
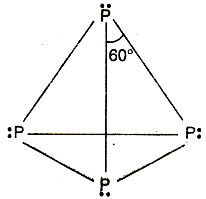
As we see in given diagram, we have
In white phosphorus ${P_4}$ ,
6 P-P single bonds
4 lone pair of electrons
Bond angle ∠P-P-P=60 degrees.
$P_4$ molecules are sometimes called as white phosphorus, in which the crystal structure of red phosphorus has a complicated network of bonding. And also, it has to be stored in water to prevent natural combustion, but red phosphorus is stable in air.
The molecular formula of white phosphorus is ${P_4}$. Four phosphorus atoms lie at the corners of the regular tetrahedron In the structure of White phosphorus. Each phosphorus atom is linked to other three phosphorus atoms by covalent bonds. The bond length of P-P is 2.21 A and PPP angle is 60 degrees.
${P_4}$ exists as tetrahedra tetratomic discrete molecule with P−P−P bond angle of 60$^\circ$,
Hence atomicity of P=4.
Note: White phosphorus is a waxy solid which burns easily and is used in chemical manufacturing and smoke munitions. When phosphorus is exposed to white may cause irritation and burns, liver, kidney, heart, lung, or bone damage, and death.
And also, it causes severely painful, partial (second degree) to full thickness (third degree) burns, which have a characteristic yellow color and garlic-like odor. burn site because Smoke may be released due to the continued burning of white phosphorus or the formation of phosphoric acid.
Complete step by step solution:

As we see in given diagram, we have
In white phosphorus ${P_4}$ ,
6 P-P single bonds
4 lone pair of electrons
Bond angle ∠P-P-P=60 degrees.
$P_4$ molecules are sometimes called as white phosphorus, in which the crystal structure of red phosphorus has a complicated network of bonding. And also, it has to be stored in water to prevent natural combustion, but red phosphorus is stable in air.
The molecular formula of white phosphorus is ${P_4}$. Four phosphorus atoms lie at the corners of the regular tetrahedron In the structure of White phosphorus. Each phosphorus atom is linked to other three phosphorus atoms by covalent bonds. The bond length of P-P is 2.21 A and PPP angle is 60 degrees.
${P_4}$ exists as tetrahedra tetratomic discrete molecule with P−P−P bond angle of 60$^\circ$,
Hence atomicity of P=4.
Note: White phosphorus is a waxy solid which burns easily and is used in chemical manufacturing and smoke munitions. When phosphorus is exposed to white may cause irritation and burns, liver, kidney, heart, lung, or bone damage, and death.
And also, it causes severely painful, partial (second degree) to full thickness (third degree) burns, which have a characteristic yellow color and garlic-like odor. burn site because Smoke may be released due to the continued burning of white phosphorus or the formation of phosphoric acid.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




