Teflon is an example of polymer which is/are -
A.Polyamide
B.Addition polymer
C.Polyester
D.Formaldehyde resin.
Answer
581.4k+ views
Hint:It is known, a polymer is a substance or material which consists of a very large number of molecules, or macromolecules, and is composed of many repeating subunits. Basically, they are materials made of long, repeating chains of molecules.
Complete step by step answer:
Polymers , based on their polymerisation, can be classified mainly into two categories -
Addition polymerisation, which is basically when a polymer is, is formed by simple linking of monomers without the co-generation of other products.
Condensation polymerisation, which is formed by the step-growth polymerization in which monomers or oligomers react with each other to form larger structural units while releasing smaller molecules as a by - product such as water or methanol, mainly water.
So as we know, Teflon is formed from the monomer tetrafluoroethene by its repeated addition. It is also a homopolymer i.e. consisting of a single monomer.
And hence option B is the correct answer.
Additional information
The structure of the monomer of Teflon is -
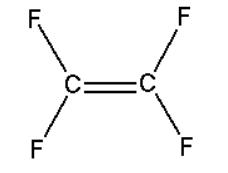
The carbons are $s{p_2}$ hybridized, and they share a double bond while the two fluorine atoms present are bonded to each carbon, and their geometry around the carbon atoms is of trigonal planar.
Note:
Polytetrafluoroethylene or Teflon is best known for its use in making the coating of non-stick frying pans and other cookware, as it is known to be hydrophobic and possesses fairly a very high heat resistance.
Complete step by step answer:
Polymers , based on their polymerisation, can be classified mainly into two categories -
Addition polymerisation, which is basically when a polymer is, is formed by simple linking of monomers without the co-generation of other products.
Condensation polymerisation, which is formed by the step-growth polymerization in which monomers or oligomers react with each other to form larger structural units while releasing smaller molecules as a by - product such as water or methanol, mainly water.
So as we know, Teflon is formed from the monomer tetrafluoroethene by its repeated addition. It is also a homopolymer i.e. consisting of a single monomer.
And hence option B is the correct answer.
Additional information
The structure of the monomer of Teflon is -

The carbons are $s{p_2}$ hybridized, and they share a double bond while the two fluorine atoms present are bonded to each carbon, and their geometry around the carbon atoms is of trigonal planar.
Note:
Polytetrafluoroethylene or Teflon is best known for its use in making the coating of non-stick frying pans and other cookware, as it is known to be hydrophobic and possesses fairly a very high heat resistance.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




