How will you synthesize?
A. Cumene from benzene
B. Benzene to n-propyl benzene
Answer
589.8k+ views
Hint: Current is defined as the rate of flow of electric charge per unit time. Water has the molecular formula \[{H_2}O\] and hence, it has two hydrogens and one oxygen. When water decomposes on passing current, it forms hydrogen and oxygen molecules.
Complete step by step answer:
The IUPAC name for cumene is (Propan2-yl) benzene. It is also known as isopropyl benzene. n-propyl benzene and isopropyl benzene are structural isomers. Structural isomers are compounds with the same molecular formula but different structural formulas.
Step-by-step answer:
(i)When benzene reacts with propylene in presence of anhydrous aluminium chloride, it forms cumene. The reaction is known as Friedel –Crafts alkylation and it can be written as,
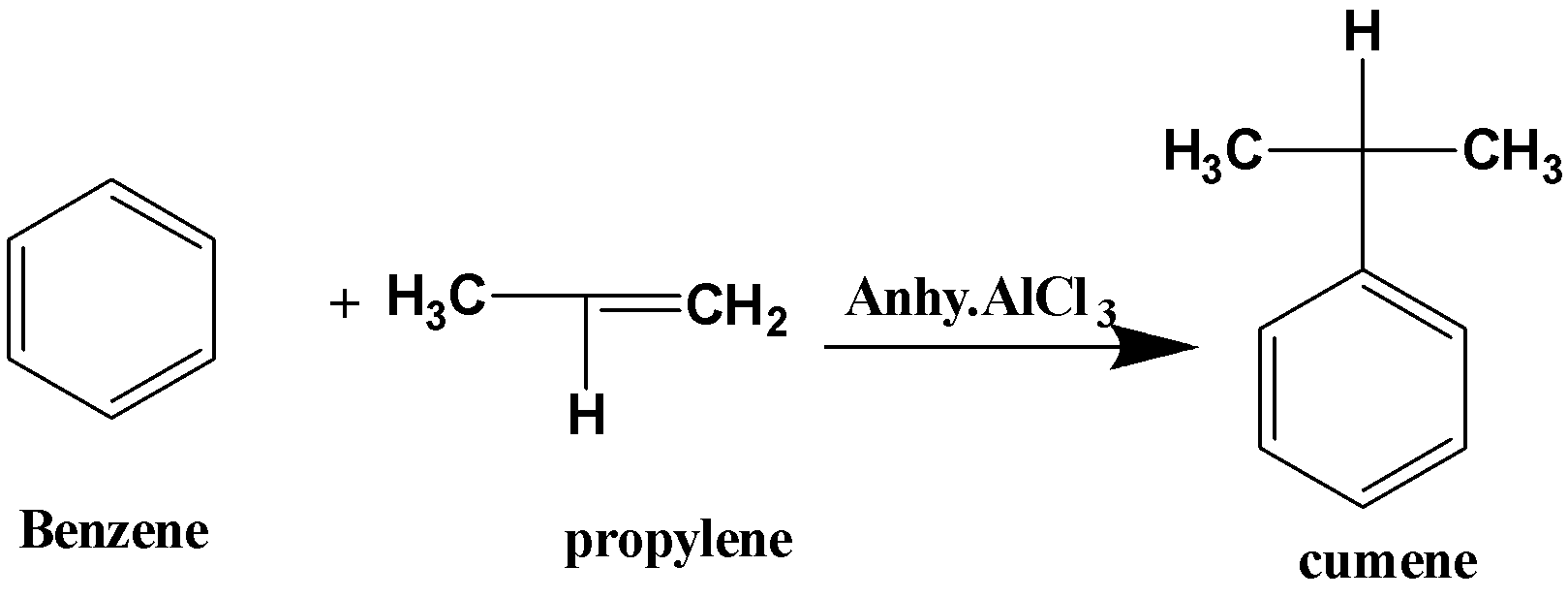
Cumene is mainly used for the preparation of organic compounds like phenol and acetone. It is used in the manufacture of rubber, iron, and paper. Cumene acts as thinner in paints and lacquers.
Benzene is the basic aromatic compound. Benzene acts as a precursor for several organic compound preparations. It is mainly used in the preparation of some dyes, pharmaceuticals drugs-related compounds.
(ii) When benzene reacts with n-propyl chloride in presence of anhydrous aluminium chloride, it forms n-propyl benzene. The reaction is known as Friedel-Crafts alkylation and it can be written as follows,
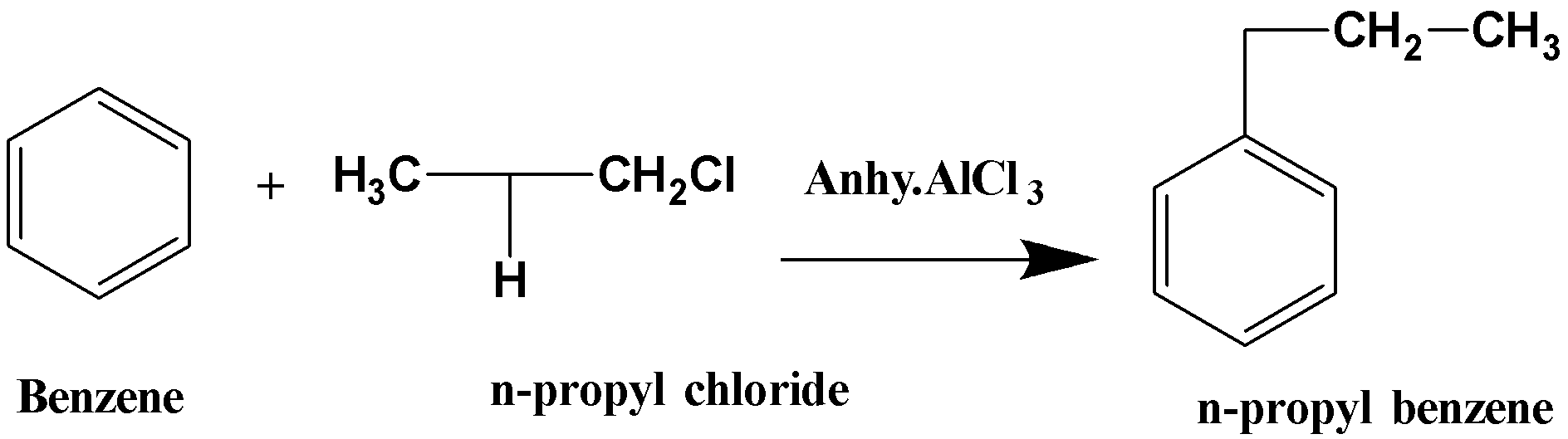
n-propyl benzene acts as non-polar solvents in the textile and printing industry. It is also used in the preparation of methyl styrene.
Note: Alternate method:
Benzene can also be converted to n-propyl benzene via Friedel-Crafts acylation followed by reduction.
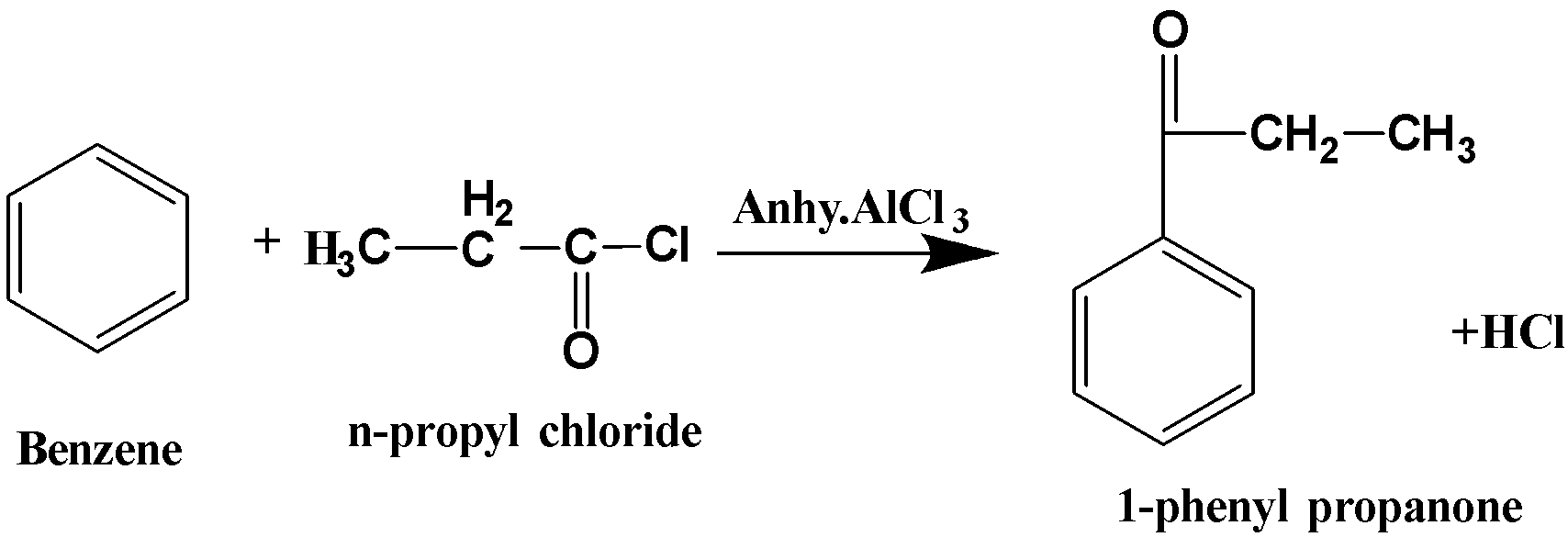
When benzene reacts with propanoyl chloride in presence of anhydrous aluminium chloride, it forms 1-phenyl propanone. The reaction is known as Friedel-Crafts acylation since the acyl group is added to the benzene. Thus, the reaction can be written as follows,
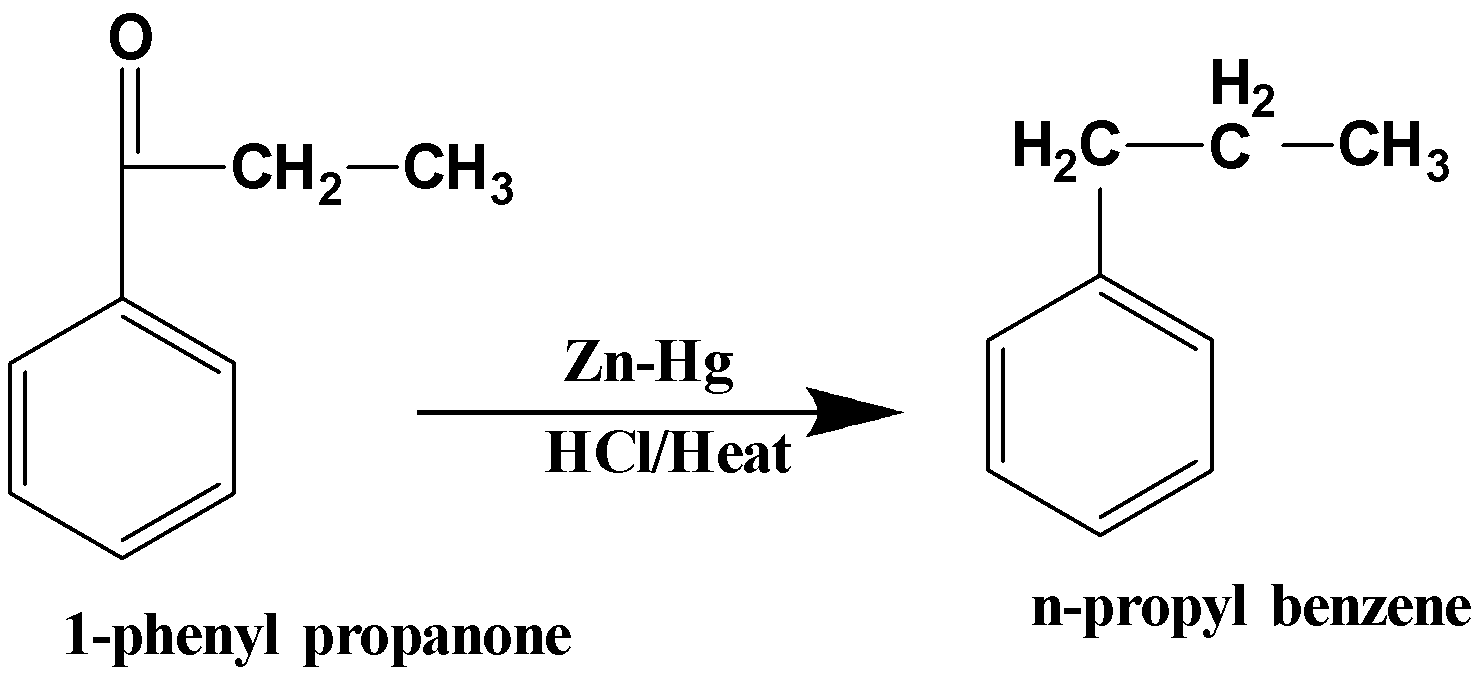
When 1-phenyl propanone is further reduced with zinc –amalgam, it forms n-propyl benzene. The reaction can be written as follows,
Complete step by step answer:
The IUPAC name for cumene is (Propan2-yl) benzene. It is also known as isopropyl benzene. n-propyl benzene and isopropyl benzene are structural isomers. Structural isomers are compounds with the same molecular formula but different structural formulas.
Step-by-step answer:
(i)When benzene reacts with propylene in presence of anhydrous aluminium chloride, it forms cumene. The reaction is known as Friedel –Crafts alkylation and it can be written as,

Cumene is mainly used for the preparation of organic compounds like phenol and acetone. It is used in the manufacture of rubber, iron, and paper. Cumene acts as thinner in paints and lacquers.
Benzene is the basic aromatic compound. Benzene acts as a precursor for several organic compound preparations. It is mainly used in the preparation of some dyes, pharmaceuticals drugs-related compounds.
(ii) When benzene reacts with n-propyl chloride in presence of anhydrous aluminium chloride, it forms n-propyl benzene. The reaction is known as Friedel-Crafts alkylation and it can be written as follows,

n-propyl benzene acts as non-polar solvents in the textile and printing industry. It is also used in the preparation of methyl styrene.
Note: Alternate method:
Benzene can also be converted to n-propyl benzene via Friedel-Crafts acylation followed by reduction.
When benzene reacts with propanoyl chloride in presence of anhydrous aluminium chloride, it forms 1-phenyl propanone. The reaction is known as Friedel-Crafts acylation since the acyl group is added to the benzene. Thus, the reaction can be written as follows,

When 1-phenyl propanone is further reduced with zinc –amalgam, it forms n-propyl benzene. The reaction can be written as follows,

Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




