What is sublimation? Perform an experiment to demonstrate the sublimation of camphor or ammonium chloride. Draw a labelled diagram.
Answer
534k+ views
Hint: Sublimation is the process of a substance transitioning directly from a solid to a gas state without passing through a liquid stage. Sublimation is an endothermic process that occurs when a substance's triple point on its phase diagram, which corresponds to the lowest pressure at which the substance can exist as a liquid, is reached. Deposition or desublimation is the reversal of sublimation, in which a substance transitions from a gas to a solid state.
Complete answer:
A solid-to-gas transition (sublimation) followed by a gas-to-solid transition has also been referred to as sublimation (deposition). While vaporisation from a liquid to a gas occurs as evaporation from the surface if it occurs below the boiling point of the liquid and as boiling with the formation of bubbles in the interior of the liquid if it occurs at the boiling point, the solid-to-gas transition occurs as sublimation from the surface.
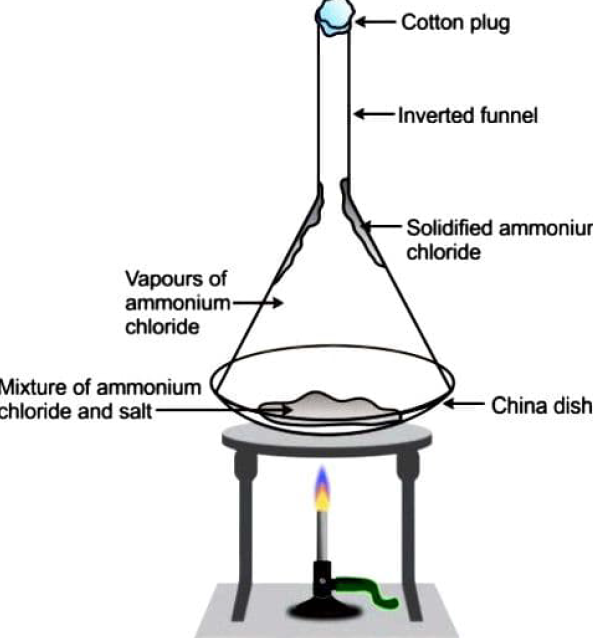
We know that when ammonium chloride is heated, it transforms from a solid to a gas. The sublimation procedure is used to separate a combination containing a sublimable volatile component from a non-sublimable contaminant (in this case, salt). It's a phase transition in which stuff travels straight from solid to gaseous form, or vapour, without going through the more typical liquid phase in between.
Note:
Most chemical compounds and elements have three distinct states at different temperatures under normal pressures. The shift from solid to gaseous state in many circumstances necessitates an intermediate liquid state. The pressure being discussed is the partial pressure of the substance, not the entire system's total (e.g. atmospheric) pressure.
Complete answer:
A solid-to-gas transition (sublimation) followed by a gas-to-solid transition has also been referred to as sublimation (deposition). While vaporisation from a liquid to a gas occurs as evaporation from the surface if it occurs below the boiling point of the liquid and as boiling with the formation of bubbles in the interior of the liquid if it occurs at the boiling point, the solid-to-gas transition occurs as sublimation from the surface.

We know that when ammonium chloride is heated, it transforms from a solid to a gas. The sublimation procedure is used to separate a combination containing a sublimable volatile component from a non-sublimable contaminant (in this case, salt). It's a phase transition in which stuff travels straight from solid to gaseous form, or vapour, without going through the more typical liquid phase in between.
Note:
Most chemical compounds and elements have three distinct states at different temperatures under normal pressures. The shift from solid to gaseous state in many circumstances necessitates an intermediate liquid state. The pressure being discussed is the partial pressure of the substance, not the entire system's total (e.g. atmospheric) pressure.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE




