How many structural isomers can you draw for pentane?
Answer
622.2k+ views
Hint: Structural isomers are those which have the same molecular formula and different bonding patterns and atomic organization. Pentane is an organic compound with 5 carbon atoms attached with single bonds. So, just see the number of arrangements formed by pentane.
Complete step by step answer:
-Pentane is a 5 carbon organic compound and since the suffix is –ane we can say that there is only single bonds (it is a saturated molecule). There are no double or triple bonds (it is not unsaturated).
-The molecular formula of Pentane is: \[{C_5}{H_{12}}\]
-The 5 carbon atoms of pentane can be arranged in following distinct forms:
n-pentane or pentane: in this first arrangement all the 5 carbon atoms are arranged one after the other to form a single chain with no branching. It is known as n-pentane and its molecular formula is \[{C_5}{H_{12}}\]. Its structural formula is:
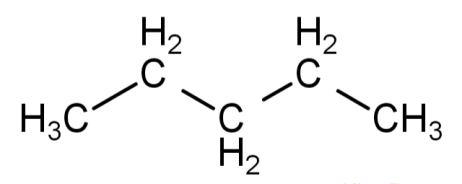
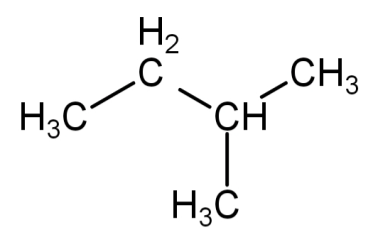
Isopentane or 2-methylbutane: in this type of arrangement 4 carbon atoms are arranged in a straight chain and the 5th carbon atom is attached to the 2nd carbon atom of the straight chain to form a branch. This is known as isopentane and the IUPAC name is 2-methylbutane. Its molecular formula is \[{C_5}{H_{12}}\].
Its structural formula is:
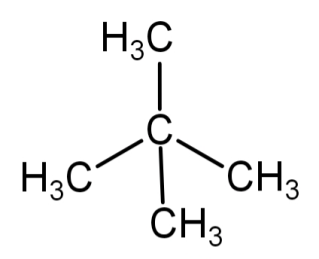
Neopentane or dimethylpropane: in this type of arrangement 3 carbon atoms are arranged one after the other to form a straight chain, while the other 2 carbon atoms are attached to the middle carbon atom of the straight chain to form 2 branches. It is known as neopentane and the IUPAC name is dimethylpropane. Its molecular formula is \[{C_5}{H_{12}}\] .
Its structural formula is:
No other patterns or types of structures are possible for pentane. So, we can say that pentane has 3 structural isomers: 1) n-pentane
2) isopentane (2-methylbutane)
3) neopentane (dimethylpropane)
Note: While naming any molecule the carbon chain should be numbered from the side where the branching is closer and not the other way round. Basically , we need to give the branch or functional group the lowest number possible. This helps us ensure that all of our isomers are distinct and not similar.
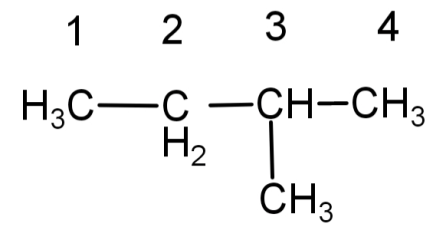
(A) Numbering done here is wrong.
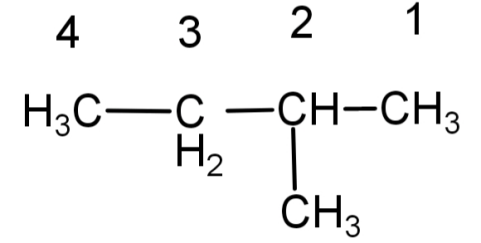
(B) Numbering done here is correct.
So, don’t confuse the above 2 isomers as different counting them as 2 isomers, they both are the same , the only difference is the numbering of the carbon atoms which is wrong in (A) and correct in (B).
Complete step by step answer:
-Pentane is a 5 carbon organic compound and since the suffix is –ane we can say that there is only single bonds (it is a saturated molecule). There are no double or triple bonds (it is not unsaturated).
-The molecular formula of Pentane is: \[{C_5}{H_{12}}\]
-The 5 carbon atoms of pentane can be arranged in following distinct forms:
n-pentane or pentane: in this first arrangement all the 5 carbon atoms are arranged one after the other to form a single chain with no branching. It is known as n-pentane and its molecular formula is \[{C_5}{H_{12}}\]. Its structural formula is:

Isopentane or 2-methylbutane: in this type of arrangement 4 carbon atoms are arranged in a straight chain and the 5th carbon atom is attached to the 2nd carbon atom of the straight chain to form a branch. This is known as isopentane and the IUPAC name is 2-methylbutane. Its molecular formula is \[{C_5}{H_{12}}\].
Its structural formula is:

Neopentane or dimethylpropane: in this type of arrangement 3 carbon atoms are arranged one after the other to form a straight chain, while the other 2 carbon atoms are attached to the middle carbon atom of the straight chain to form 2 branches. It is known as neopentane and the IUPAC name is dimethylpropane. Its molecular formula is \[{C_5}{H_{12}}\] .
Its structural formula is:

No other patterns or types of structures are possible for pentane. So, we can say that pentane has 3 structural isomers: 1) n-pentane
2) isopentane (2-methylbutane)
3) neopentane (dimethylpropane)
Note: While naming any molecule the carbon chain should be numbered from the side where the branching is closer and not the other way round. Basically , we need to give the branch or functional group the lowest number possible. This helps us ensure that all of our isomers are distinct and not similar.

(A) Numbering done here is wrong.

(B) Numbering done here is correct.
So, don’t confuse the above 2 isomers as different counting them as 2 isomers, they both are the same , the only difference is the numbering of the carbon atoms which is wrong in (A) and correct in (B).
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which Indian city is known as the "City of Victory"?

Which instrument is used to measure the Blood Pressure?

Distinguish between the reserved forests and protected class 10 biology CBSE

Fill the blanks with proper collective nouns 1 A of class 10 english CBSE

Fill in the blank using the plural form of the noun class 10 english CBSE

What is resource planning Why is resource planning class 10 social science CBSE




