What is the structural formula of cyclobutane?
Answer
559.5k+ views
Hint: From the name of the compound, we can infer that
- It has a cyclic structure
- It has four carbon atoms
- It does not have any multiple bonds
Complete answer:
We know that cyclobutane is a type of cycloalkane. A cycloalkane is a monocyclic saturated hydrocarbon. It does not contain any atoms or elements except hydrogen and carbon.
Since we know that it has a saturated monocyclic structure and has four carbon atoms, its Lewis structure will be as follows
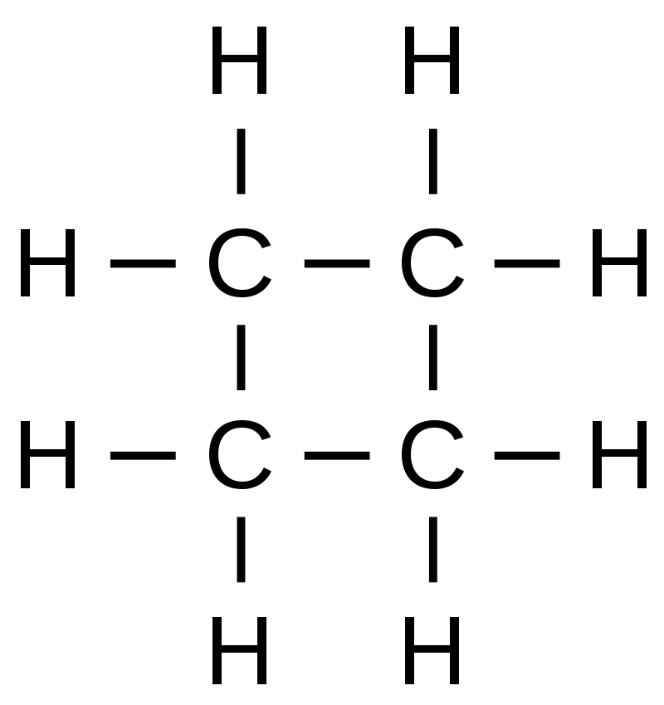
So, from the structure, we can see that it has 8 hydrogen atoms and hence structural formula of cyclobutane will be ${{(C{{H}_{2}})}_{4}}$ or ${{C}_{4}}{{H}_{8}}$.
Additional Information:
Some properties of cyclobutane are
- It is prepared by dimerization of alkenes in UV-light. It can also be prepared by dehalogenation of 1,4-dihalobutene in the presence of reducing metals.
- It is a colorless gas but can be liquified for commercial purposes.
- It has a molar mass of 56.107 g/mol.
- Its boiling point is 285.6K and its melting point is 182K.
- Complex derivatives of cyclobutane are of much importance in the fields of biotechnology and biology.
- It does not have much biological or commercial significance.
- Carbon-carbon bonds in cyclobutane have lower bond energy and have significantly strained bond angles. This is why it is unstable at a temperature above $500{}^\circ C$.
Note:
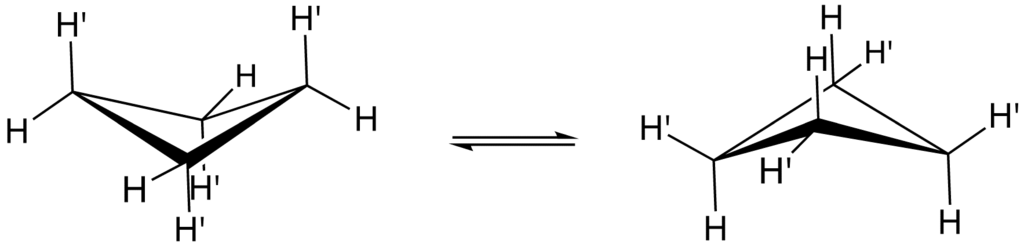
It should be noted that the four carbon atoms in the cyclobutane compound are not coplanar. A puckered or a folded conformation is adopted by the cyclobutane ring. The angle between the plane formed by the three carbon atoms and the fourth carbon atom is $25{}^\circ $.
- It has a cyclic structure
- It has four carbon atoms
- It does not have any multiple bonds
Complete answer:
We know that cyclobutane is a type of cycloalkane. A cycloalkane is a monocyclic saturated hydrocarbon. It does not contain any atoms or elements except hydrogen and carbon.
Since we know that it has a saturated monocyclic structure and has four carbon atoms, its Lewis structure will be as follows

So, from the structure, we can see that it has 8 hydrogen atoms and hence structural formula of cyclobutane will be ${{(C{{H}_{2}})}_{4}}$ or ${{C}_{4}}{{H}_{8}}$.
Additional Information:
Some properties of cyclobutane are
- It is prepared by dimerization of alkenes in UV-light. It can also be prepared by dehalogenation of 1,4-dihalobutene in the presence of reducing metals.
- It is a colorless gas but can be liquified for commercial purposes.
- It has a molar mass of 56.107 g/mol.
- Its boiling point is 285.6K and its melting point is 182K.
- Complex derivatives of cyclobutane are of much importance in the fields of biotechnology and biology.
- It does not have much biological or commercial significance.
- Carbon-carbon bonds in cyclobutane have lower bond energy and have significantly strained bond angles. This is why it is unstable at a temperature above $500{}^\circ C$.
Note:
It should be noted that the four carbon atoms in the cyclobutane compound are not coplanar. A puckered or a folded conformation is adopted by the cyclobutane ring. The angle between the plane formed by the three carbon atoms and the fourth carbon atom is $25{}^\circ $.

Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE




