Sodium is a very reactive metal. Is this statement true or false?
Answer
537.6k+ views
Hint: The reactivity of an element is dependent on a lot of factors like its size, the ability to lose or gain electrons and the effective nuclear charge experienced by the valence electrons of the atom. A series of reactions and their observations has led to the formation of a reactivity series.
Complete answer:
Sodium is an alkali metal belonging to the first group of the periodic table. All alkali metals have a single electron in their valence shells. The considerably large sizes of alkali metals reduces the effective nuclear charge experienced by the valence electron which is responsible for their low ionization energies and high electropositivity.
A metal is said to be reactive if it is highly electropositive in nature and readily loses its electrons in a reaction. Sodium metal has very low ionization energy and therefore readily loses its electron.
\[Na(g) \to N{a^ + }(g) + {e^ - }{\text{ I}}{\text{.E = 496}}KJmo{l^{ - 1}}\]
Sodium reacts vigorously with oxygen and water releasing enormous amounts of energy (highly exothermic reactions) that it catches fire and explodes with popping sounds. Therefore it is always stored in kerosene as even little exposure results in hazardous reactions.
\[2Na(s) + 2{H_2}O(l) \to 2NaOH(aq) + {H_2}(g) + {\text{large amount of heat energy}}\]
Such intense reactions with water, air and acids shown by sodium make it the second most reactive metal in the reactivity series.
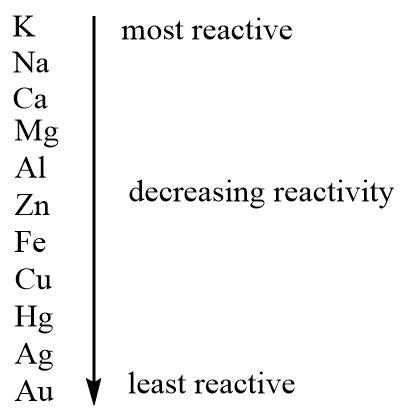
Hence it is true that sodium is a very reactive metal.
Note:
The reaction for ionization was written in the gas phase even though sodium is metal that exists in solid state because ionization enthalpy is always calculated when an element is in its ground state, gaseous form and completely isolated from other atoms or molecules.
Complete answer:
Sodium is an alkali metal belonging to the first group of the periodic table. All alkali metals have a single electron in their valence shells. The considerably large sizes of alkali metals reduces the effective nuclear charge experienced by the valence electron which is responsible for their low ionization energies and high electropositivity.
A metal is said to be reactive if it is highly electropositive in nature and readily loses its electrons in a reaction. Sodium metal has very low ionization energy and therefore readily loses its electron.
\[Na(g) \to N{a^ + }(g) + {e^ - }{\text{ I}}{\text{.E = 496}}KJmo{l^{ - 1}}\]
Sodium reacts vigorously with oxygen and water releasing enormous amounts of energy (highly exothermic reactions) that it catches fire and explodes with popping sounds. Therefore it is always stored in kerosene as even little exposure results in hazardous reactions.
\[2Na(s) + 2{H_2}O(l) \to 2NaOH(aq) + {H_2}(g) + {\text{large amount of heat energy}}\]
Such intense reactions with water, air and acids shown by sodium make it the second most reactive metal in the reactivity series.

Hence it is true that sodium is a very reactive metal.
Note:
The reaction for ionization was written in the gas phase even though sodium is metal that exists in solid state because ionization enthalpy is always calculated when an element is in its ground state, gaseous form and completely isolated from other atoms or molecules.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




